Theme 1: Rational Conformational Control
Current and recent funding in this area – EPSRC Programme Grant ‘Molecular Robotics’; European Research Council Advanced Grant DOGMATRON, Leverhulme Trust, EPSRC studentships.
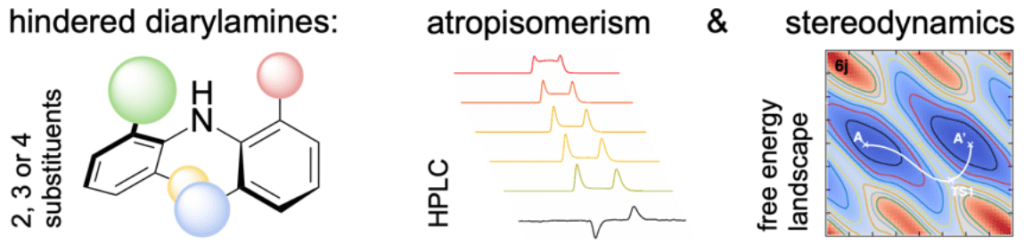
1.1 Atropisomers: asymmetric synthesis and dynamics
Atropisomers—compounds which are chiral by virtue of restricted rotation about a single bond—have provided chemists with some of the most successful ever ligands for asymmetric catalysis. Our research in this area addresses two areas where there are major opportunities for development.
a) Discovery and application of new atropisomer classes
The vast majority of atropisomeric structures fall into a single class: biaryls, and almost all involve rotational restriction about a C–C bond. Over the last 15 years we have been exploring atropisomers based on unconventional structures involving amides, ethers, sulfur compounds and ureas. We have discovered some mechanistically intriguing ways of making them as single enantiomers using dynamic resolution techniques, and we have explored their application as new classes of chiral ligands. Atropisomers are in effect ‘frozen conformers’, and our work has made extensive use of the connections between conformational control and atropisomerism. We’re now looking to use the methods and systems we have pioneered to build new classes of atropisomeric ligands for use in asymmetric catalysis.
b) Methods for the asymmetric synthesis of atropisomers
Even well established atropisomer classes are not typically made by asymmetric synthesis: for some, tedious resolution methods are required. We are investigating alternative methods involving dynamic kinetic resolution or dynamic thermodynamic resolution for achieving atroposelective synthesis. Recently for example we have explored the application of artificially evolved enzymes catalyzing redox processes to the asymmetric synthesis of atropisomers of potential value in the synthesis of chiral ligands for asymmetric synthesis. Atropisomeric drugs are also of increasing importance in medicinal chemistry (see our review at Angew. Chemie Int. Ed. 2009, 48, 6398-6401).
c) Dynamics of atropisomers and ‘near-atropisomers’
Low-barrier biaryls and other hindered systems that exist as slowly interconverting conformers are not technically atropisomers, but she many features in terms of synthesis and dynamics. We are currently developing new molecular components – including motors and gear trains – based around these structures.
Recent work in this area:
Atropisomerism in diarylamines: structural requirements and mechanisms of conformational interconversion, Romain Costil, Alistair Stirling, Fernanda Duarte and Jonathan Clayden, Angew. Chemie. Int. Ed. in press [doi 10.1002/anie.202007595]
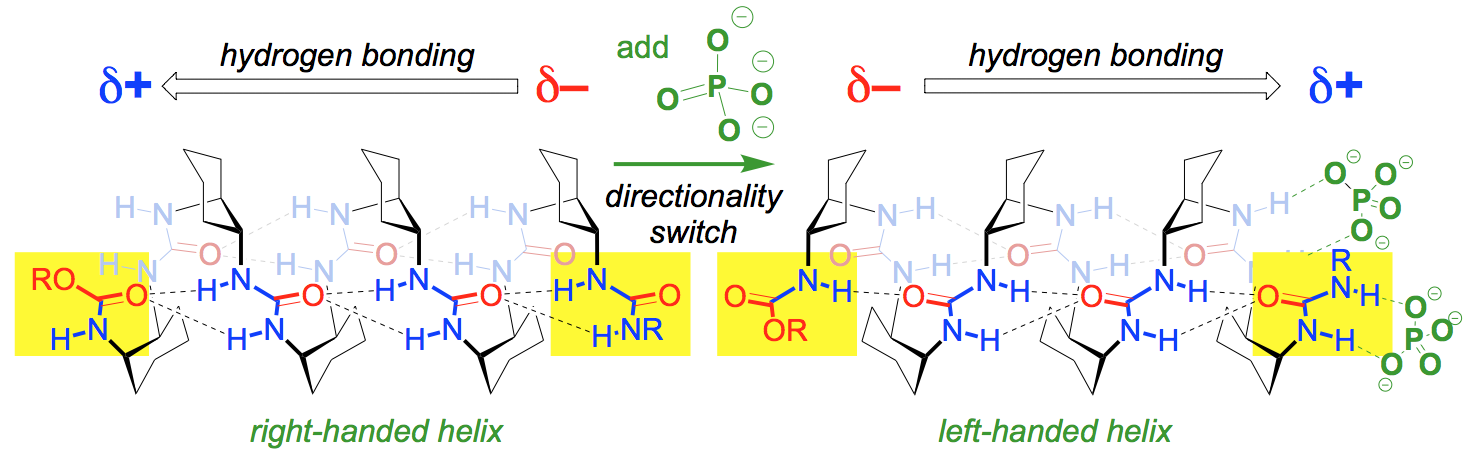
1.2 Dynamic Foldamer Chemistry
Helical structures are ubiquitous in Nature, and synthetic structures with well-defined helical conformations are termed ‘foldamers’. We have explored separately the concepts of helical preference (a thermodynamic preference for a global helical structure, as opposed to a random coil) and screw-sense preference (the thermodynamic preference for that helix to adopt a specific left- or right-handedness), by building synthetic helices made from achiral monomers. In biomolecular helices, these concepts are entangled because the chiral monomers (for example amino acids or nucleosides) making up the helix enforce a preference for both helicity and screw-sense. Using some simple (but previously overlooked) spectroscopic and stereochemical principles, we have developed NMR (1H, 13C and 19F) methods, probes and markers for exploring the kinetics and thermodynamics of helical interconversions on a range of timescales. We are now extending these probes to include more sensitive fluorescent and coloured reporters. We are also interested in stressed helices – helices with faults, bends and breaks. These have rarely been explored, but throw light on mechanisms of helical interconversion in foldamer structures. By forcing a helix to contain a fault structure, we hope to characterize in detail unusual conformational features such as the elusive peptide gamma turn.
Recent work in this area:
Competing hydrogen-bond polarities in a dynamic oligourea foldamer: a molecular spring torsion balance, Romina Wechsel, Matej Žabka, John W. Ward and Jonathan Clayden, J. Am. Chem. Soc. 2018, 140, 3528-3531 [doi 10.1021/jacs.8b00567]
1.3 Molecular Communication Devices
Nature uses molecular conformation in subtle and sometimes elaborate ways to control function and to communicate information—allosteric effects in enzymes, or switching in receptors, for example.
a) Ultra-remote stereocontrol
We have constructed extended molecules in which conformational preferences are relayed over unprecedentedly long distances, and have for the last 10 years been the world record holders for remote stereocontrol (1,23-asymmetric induction published in 2004; 1, 31-, 1,46- and 1,61-asymmetric induction published in 2013). Current work is seeking to extend the ability to control reactions at a distance to catalytic systems, which would allow us to make even more biomimetic receptor mimics with chemical, rather than spectroscopic, outputs. We are also looking to achieve remote stereocontrol through polymers – in other words extend the distances over which information is communicated beyond the nanoscale towards the 0.01-1 micrometre scale.
Representative work in this area:
Foldamer-mediated remote stereocontrol: >1,60 asymmetric induction, Liam Byrne, Jordi Solà, Thomas Boddaert, Tommaso Marcelli, Ralph W. Adams, Gareth A. Morris and Jonathan Clayden, Angew. Chemie. Int. Ed. 2014, 53, 151-155 [doi 10.1002/anie.201308264] (‘VIP paper’)

See also Highlights summary article: C. P. Johnston and M. D. Smith Angew. Chem. Int. Ed. 2014, 53, 3315-3317 [doi 10.1002/anie.201400154]
b) Synthesis of membrane-bound artificial receptors.
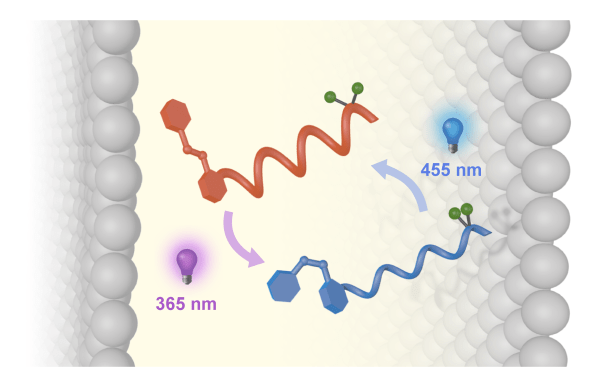
We are currently working to make molecules which mimic biology’s ability to encode, manipulate and communicate information. We have reported artificial receptors that bind ligands and relay information about their structure and stereochemistry to a spectroscopic reporter lying some nanometre distances away. We have recently used these molecules in systems that function in the membrane environment, potentially allowing us to communicate information across lipid bilayers, in a manner reminiscent of the G-protein coupled receptor. The protein rhodopsin, central to the function of human vision, is a modified receptor, and we have reported an artificial rhodopsin mimics that captures information about incident light and communicate that information to a distance chemical site through a conformational change.
Representative publications in this area:
Ligand-modulated conformational switching in a fully synthetic membrane-bound receptor, Francis G. A. Lister, Bryden A. F. Le Bailly, Simon J. Webb and Jonathan Clayden, Nature Chem., 2017, 9, 420-425 [doi 10.1038/nchem.2736]. See also the Nature Chemistry editorial ‘Sending a message to the other side’ (Nature Chem., 2017, 9, 403 [doi 10.1038/nchem.2776]) and Highlight article ‘Membrane messengers’ (Nature Chem., 2017, 9, 403 [doi 10.1038/nchem.2775]) covering this work.
Conformational photoswitching of a synthetic peptide foldamer bound within a phospholipid bilayer, Matteo De Poli, Wojciech Zawodny, Ophélie Quinonero, Mark Lorch, Simon J. Webb and Jonathan Clayden, Science 2016, 352, 575-580 [doi 10.1126/science.aad8352]
Theme 2: New Molecular Reactivity
Current and recent funding in this area – EPSRC, Syngenta, AstraZeneca, GlaxoSmithKline, EPSRC studentships
Over the last 20 years we have discovered ways in which the restricted conformation of planar functional groups such as amides and ureas endue them, and their anionic derivatives, with remarkable new reactivity. Many of these reactions fly in the face of well-established principles of polar reactivity: for example, we discovered how to force anions to attack electron-rich aromatic rings and alkenes, as well as new ways to destroy aromaticity. We have used the resulting partially saturated products as starting materials in a variety of synthetic strategies.
2.1 Carbolithiation of electron-rich π systems: stereoselective arylation, vinylation and alkylation methods
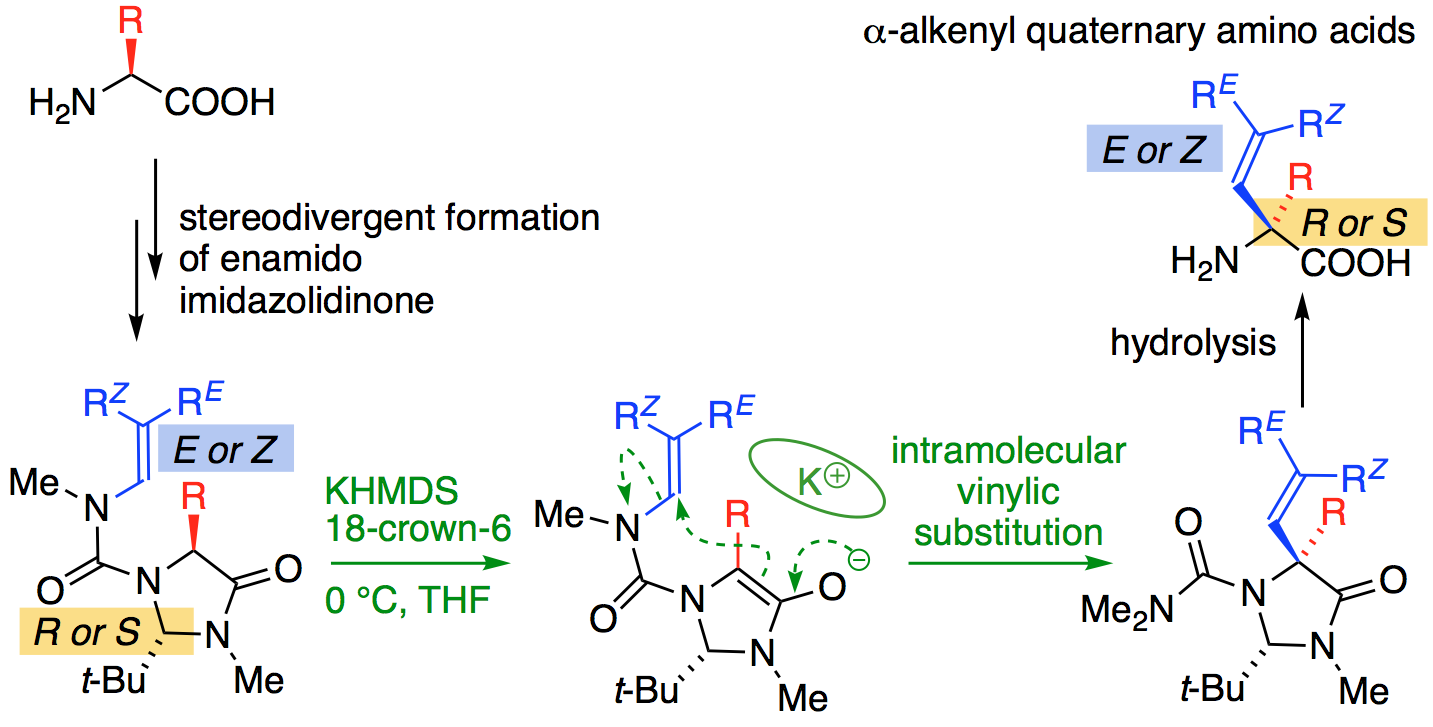
We are currently exploring ways in which the geometry of te urea linkage and its congeners allows the formation of C–C bonds by unusual, often stereospecific, reactions. One target group of interest at present are quaternary amino acids arylated at their a-position, which we make by an N to C aryl migration. We are exploring similar chemistry involving vinylation, which opens up the possibility of making spirocyclic compounds by metathesis chemistry. The methods we develop are then applied to the synthesis of valuable, biologically active targets.
Representative work in this area:
Asymmetric alpha-Arylation of Amino Acids, Daniel J. Leonard, John W. Ward and Jonathan Clayden, Nature 2018, 562, 105-109 [doi 10.1038/s41586-018-0553-9]
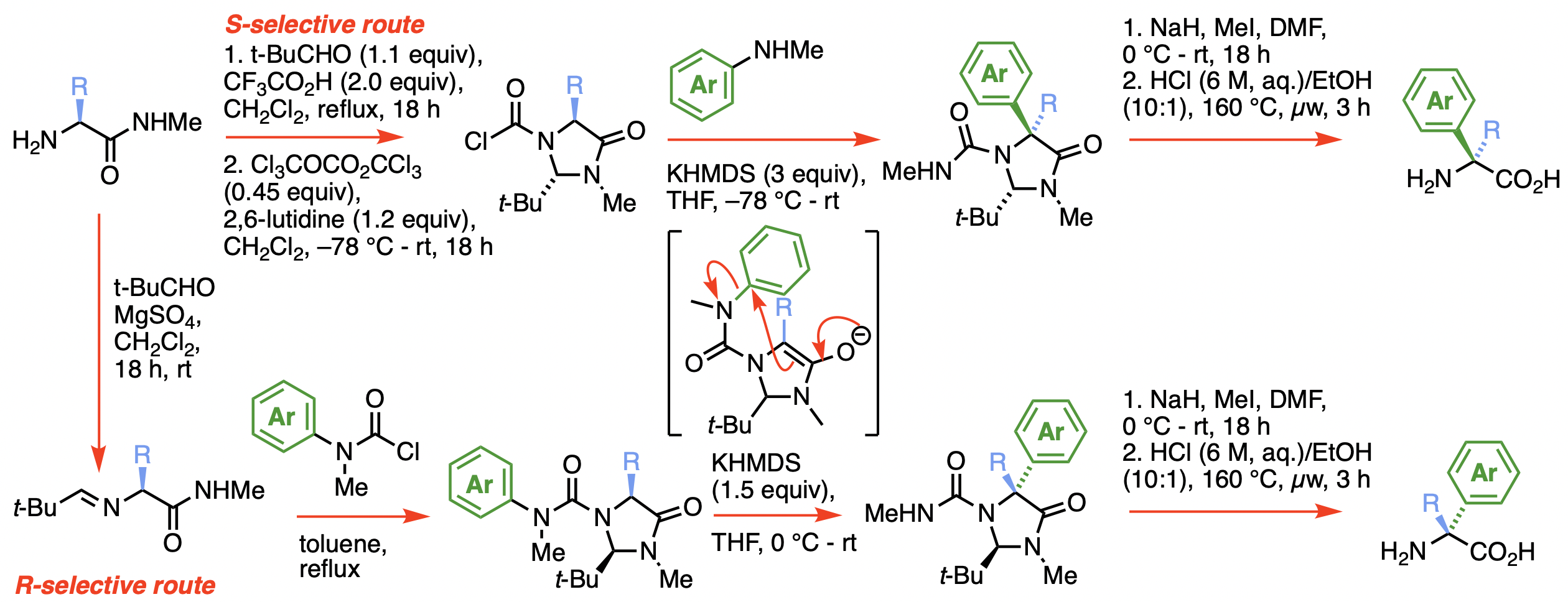
Asymmetric and Geometry-selective α-Alkenylation of α-Amino Acids, Hossay Abas, Josep Mas-Roselló, Mostafa M. Amer, Derek J. Durand, Robin R. Groleau, Natalie Fey, Jonathan Clayden Angew. Chem. Int. Ed. 2019, 58, 2418-2422 [doi 10.1002/anie.201813984]
2.2 Nucleophilic dearomatisation
We have reported several new methods for nucleophilic attack on aromatic rings that lead to non-aromatic products. Often, the remaining unsaturation provides a valuable handle for introducing further functionality, and we have used this as a strategy for the synthesis of bioactive targets including kainic acid, an acromelic acid analogue, and several members of the isodomoic acid family. Currently we are seeking to extend our methods to the dearomatising cyclisations and spirocyclisations of heterocyclic substrates.
2.3 Organolithium structure, reactivity and photochemistry
Organolithiums are the bedrock of many synthetic methods, combining predictable selectivity with broad reactivity. Many of our synthetic methods are based on organolithium chemistry, and we are exploring the links between conformation, configuration, reactivity and selectivity in these structures. Photochemistry offers further possibilities in the area, and current work is exploring the potential for new reactions based on light-promoted reactions of organolithium.
Theme 3: Synthesis of Bioactive Compounds
Current and recent funding in this area – Alzeim Ltd., AstraZeneca, EPSRC studentships.
3.1 Medium ring, polycyclic, and partially saturated heterocycles
Medium-sized rings are hard to make because of transannular ring strain, yet medium ring heterocycles have many promising conformational features as medicinal chemistry targets. We have been developing novel ring expansion chemistry as a means of making new classes of medium ring structures. We are also using dearomatisation chemistry to build complexity from simple precursors.
Representative publications in this area:
Medium-ring analogues of dibenzodiazepines by conformationally induced Smiles ring expansion, Romain Costil, Quentin Lefebvre, Jonathan Clayden, Angew. Chemie Int. Ed. 2017, 56, 14602–14606 [doi 10.1002/anie.201708991]

Medium Ring Nitrogen Heterocycles by Migratory Ring Expansion of Metallated Ureas, Jessica E. Hall, Johnathan V. Matlock, John W. Ward, Katharine V. Gray, Jonathan Clayden, Angew. Chemie Int. Ed., 2016, 55, 11153-11157 [doi 10.1002/anie.201605714]

3.2 Isodomoic acids and their analogues – the Amnesic Shellfish Toxins
These toxins have important biological activity that reveals the details of nerve function and provides a tool for the study of Alzheimer’s and Huntington’s diseases. We have published syntheses of the pharmacologically interesting isodomoic acids, and we are seeking to extend this work to other nitrogen-containing bioactive targets.
Representative publications in this area:
A general synthetic approach to the amnesic shellfish toxins: total synthesis of (–)-isodomoic acid B, (–)-isodomoic acid E and (–)-isodomoic acid F, Gilles Lemière, Simon Sedehizadeh, Julie Toueg, Nadia Fleary-Roberts and Jonathan Clayden, Chem. Commun. 2011, 3745-3747 [doi 10.1039/C1CC00048A]

The synthesis of (–)-isodomoic acid C, Jonathan Clayden, Faye. E. Knowles and Ian R. Baldwin, J. Am. Chem. Soc. 2005, 127, 2412-2413 [doi 10.1021/ja042415g]

3.3 Arogenate chemistry and the synthesis of cyclo-modified amino acids
Arogenate is a key plant metabolite which has been made suynthetically only by relatuvely inefficient routes. We are developing a practical scalable route to arogenate and its analogues, as well as practical methods for the synthesis of other modified amino acids of potential utility in medicinal chemistry or plant science / agroscience.