2026
ii. [Preprint] Hydrogen Bonding within Dynamic 19F-Tagged Oligothiourea Foldamers in Solution and in Membranes
Lucia Trevisan, Kathryn S. Foster and Jonathan Clayden
ChemRXiv 13 Mar 2026 10.26434/chemrxiv.15000888/v1
i. [Preprint] Transmission of autonomous redox-driven directional bond rotation through a molecular gear
Theo H N Bulless, Michael G Howlett, Nicholas T O’Donoghue, Matej Zabka, Beatrice S L Collins, and Jonathan Clayden
ChemRXiv 4 Dec 2025 10.26434/chemrxiv-2025-hmnb4
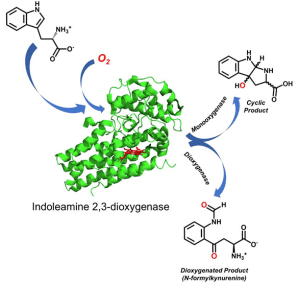
329. Monooxygenase Activity of Indoleamine 2,3-Dioxygenase
Ali Lubis, Anna Bailey, Marko Hanzevacki, Christopher Williams, Mehul Jesani, Lola González-Sánchez, Christopher Arthur, Hannah Wilson, Andrea Gallio, Peter Moody, Matthew Crump, Adrian Mulholland, Allen M. Orville, Jonathan Clayden, Emma Raven
J. Am. Chem. Soc. 2026, 148, 6178-6187 [10.1021/jacs.5c17552]
2025
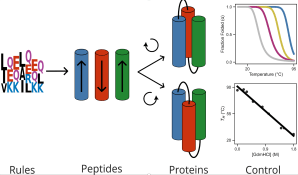
328. De novo designed 3-helix bundle peptides and proteins with controlled topology and stability
Xiyue Leng, Katherine I. Albanese, Lia R. Golub, Arthur A. Norman, Jonathan Clayden, and Derek N. Woolfson
Chemical Science 2025, 16, 18632-18641 [10.1039/D5SC05576H]
327. Redox-powered autonomous directional C–C bond rotation under enzyme control
Jordan Berreur, Olivia F. B. Watts, Theo H. N. Bulless, Nicholas T. O’Donoghue, Marc Del Olmo, Ashley J. Winter, Jonathan Clayden, and Beatrice S. L. Collins
Nature, 2025, 644, 96-101 [10.1038/s41586-025-09291-6]
View a Youtube animation of the motor in action (credit: Anna Tanczos, SciCommStudios)
Also featured in Chemistry and Engineering News
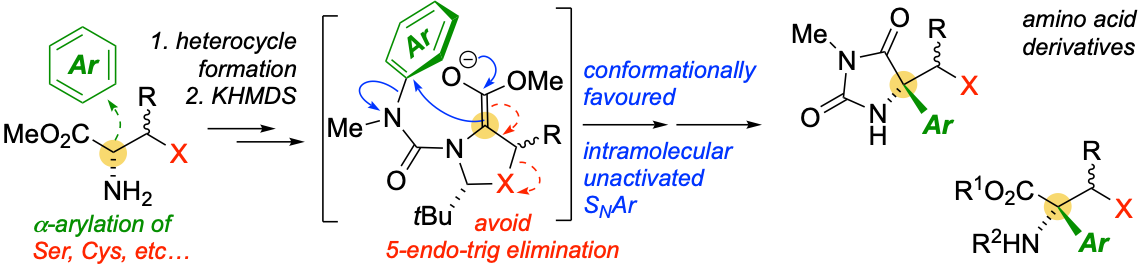
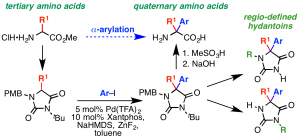
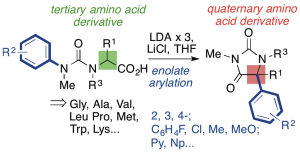
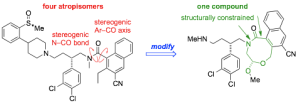
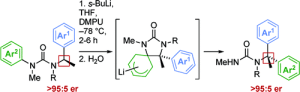
326. Asymmetric Intramolecular alpha-Arylation of Polar Amino Acids Bearing beta Leaving Groups
Ömer Taşpınar, Daniel J. Leonard, Nathan Picois, Cornelia Göcke, Matej Žabka, Hazel A. Sparkes, and Jonathan Clayden
Angew. Chemie Int. Ed. 2025, e202507713 [10.1002/anie.202507713]
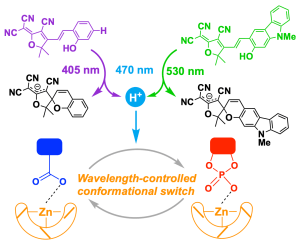
325. Using Colour to Control Conformation in a Chemical System Containing Multiple Tricyanofuran Photoacids
2024
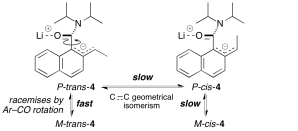
324. Intramolecular Nucleophilic Vinylic Substitution (SNV) by Carbon Nucleophiles: Conformationally Directed Formation of Dienes from N,N’-Diallyl Ureas
Branca C. van Veen and Jonathan Clayden
Chem. Eur. J. 2024, e202402352 [10.1002/chem.202402352]
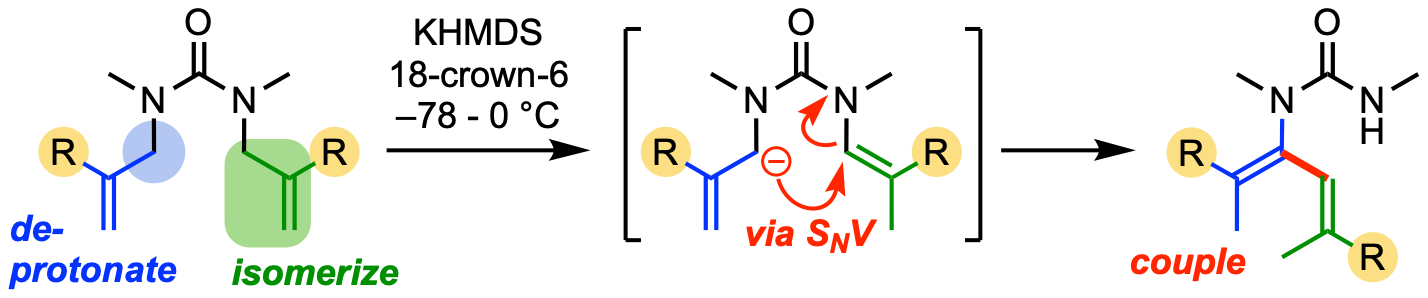
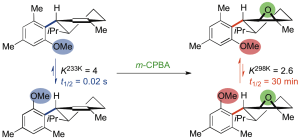
323. Selective defluorination of trifluoromethyl substituents by conformationally induced remote substitution
Mehul H. Jesani, Maria Schwarz, Shiwhu Kim, Finlay L. Evans, Alexander White, Alex Browning, Roman Abrams and Jonathan Clayden
Angew. Chem. Int. Ed. 2024, e202403477 [10.1002/anie.202403477]
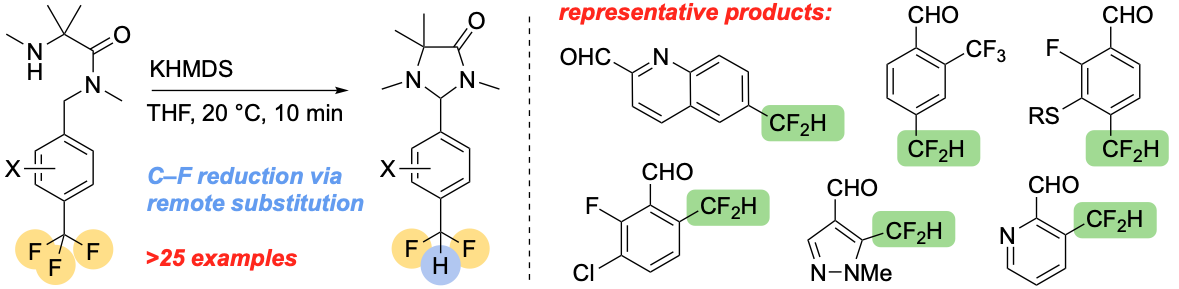
322. Benzo-fused Nitrogen Heterocycles by Asymmetric Ring Expansion and Stereochemically Retentive Re-contraction of Cyclic Ureas
Rajendra K. Mallick, Matej Žabka, and Jonathan Clayden
Angew. Chem. Int. Ed. 2024, e202318417 [10.1002/anie.202318417]
2023
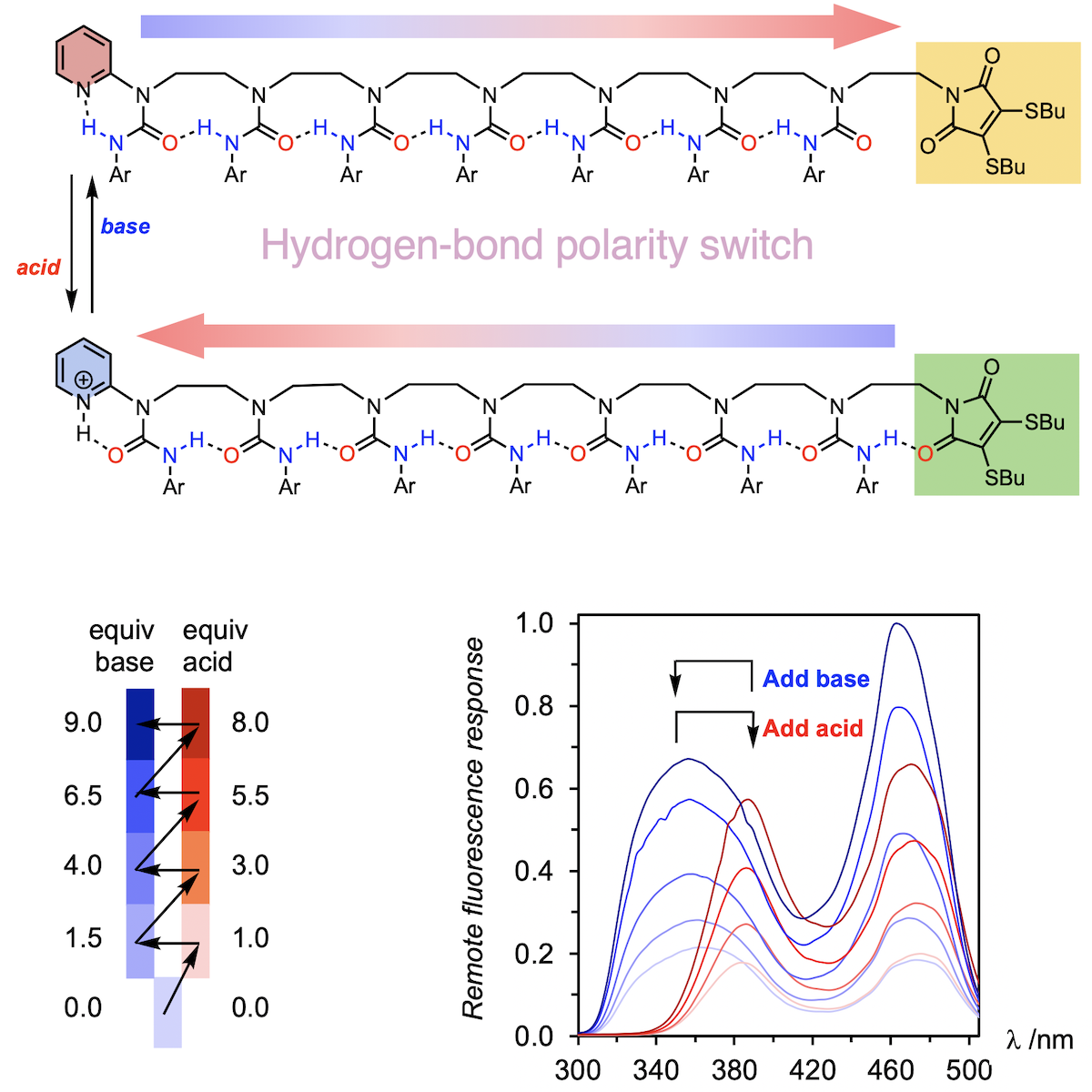
321. Anion-Dependent Hydrogen-Bond Polarity Switching in Ethylene-bridged Urea Oligomers
David P. Tilly, David T. J. Morris and Jonathan Clayden
Chem. Eur. J. 2023, e202302210 [10.1002/chem.202302210]
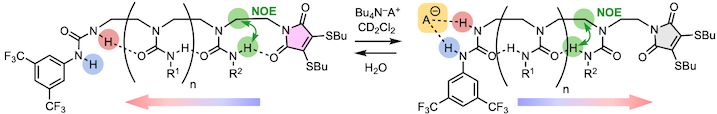
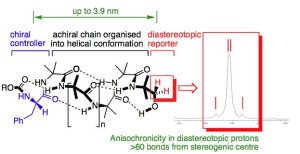
320. Dynamic and Persistent Cyclochirality in Hydrogen-Bonded Derivatives of Medium-Ring Triamines
David T. J. Morris, Steven M. Wales, Javier Echavarren, Matej Žabka, Giulia Marsico, John W. Ward, Natalie E. Pridmore and Jonathan Clayden
J. Am. Chem. Soc. 2023, 145, 19030–19041 [10.1021/jacs.3c06570]

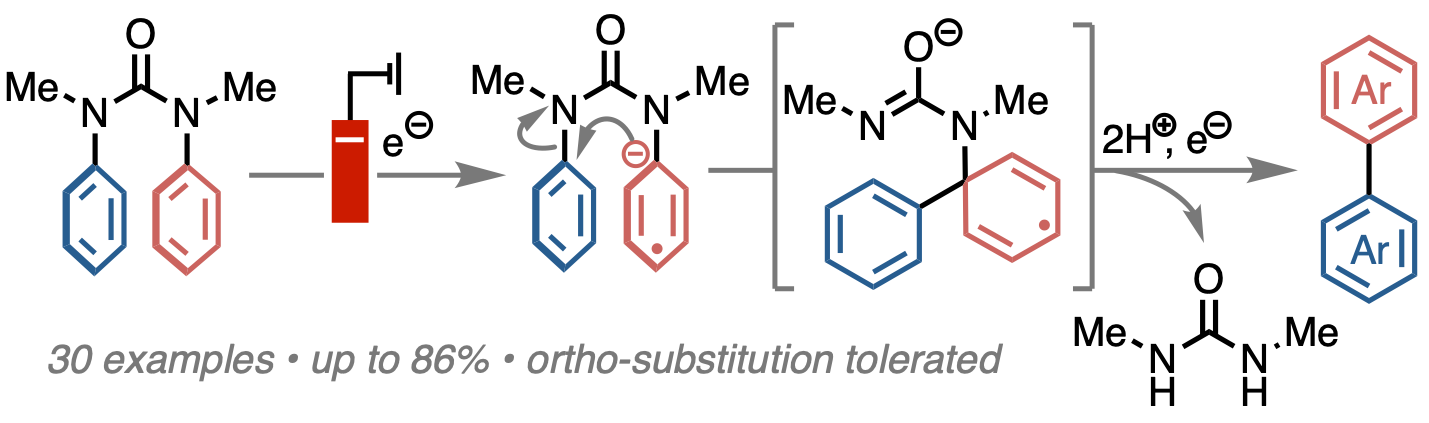
319. Electrochemical Synthesis of Biaryls by Reductive Extrusion from N,N’-Diarylureas
Ellie Stammers, Chris D. Parsons, Alastair J. J. Lennox, and Jonathan Clayden
Nature Communications, 2023, 14, 4561 [10.1038/s41467-023-40237-6]
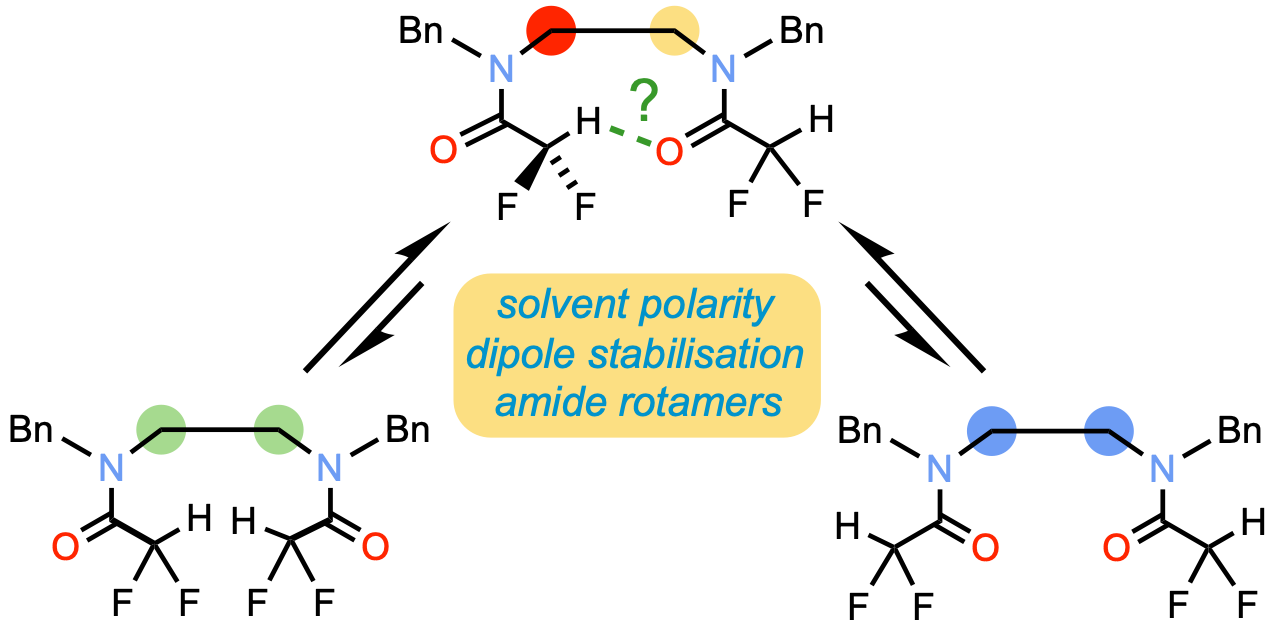
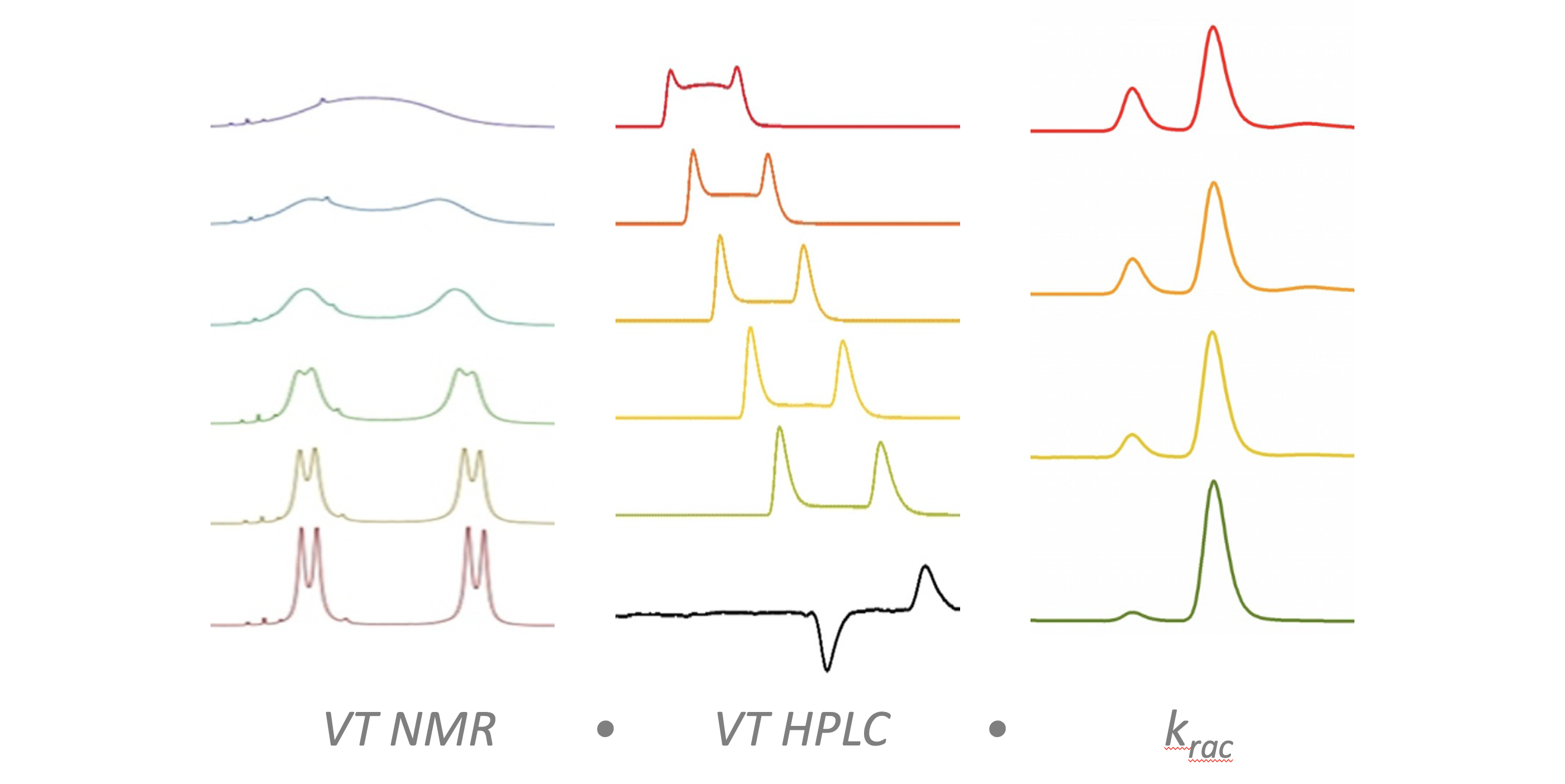
318. Conformational Preference in Difluoroacetamide Oligomers: Probing the Potential for Foldamers with C-H···O Hydrogen Bonds
Matej Žabka and Jonathan Clayden
Org. Biomol. Chem., 2023, 21, 5939-5943 [doi 10.1039/D3OB00811H]
317. Interrogating the configurational stability of atropisomers.
Jean-Paul Heeb, Jonathan Clayden, Martin D. Smith, Roly J. Armstrong
Nature Protocols, 2023, 18, 2745–2771 [10.1038/s41596-023-00859-y]
316. Switching imidazole reactivity by dynamic control of tautomer state in an allosteric foldamer
David P. Tilly, Jean-Paul Heeb, Simon J. Webb and Jonathan Clayden
Nature Communications, 2023, 14, 2647 [doi 10.1038/s41467-023-38339-2]
315. Screw Sense and Screw Sensibility: Communicating Information by Conformational Switching in Helical Oligomers
David T. J. Morris and Jonathan Clayden
Chem. Șoc. Rev. 2023, 52, 2480-2496 [doi 10.1039/D2CS00982J]

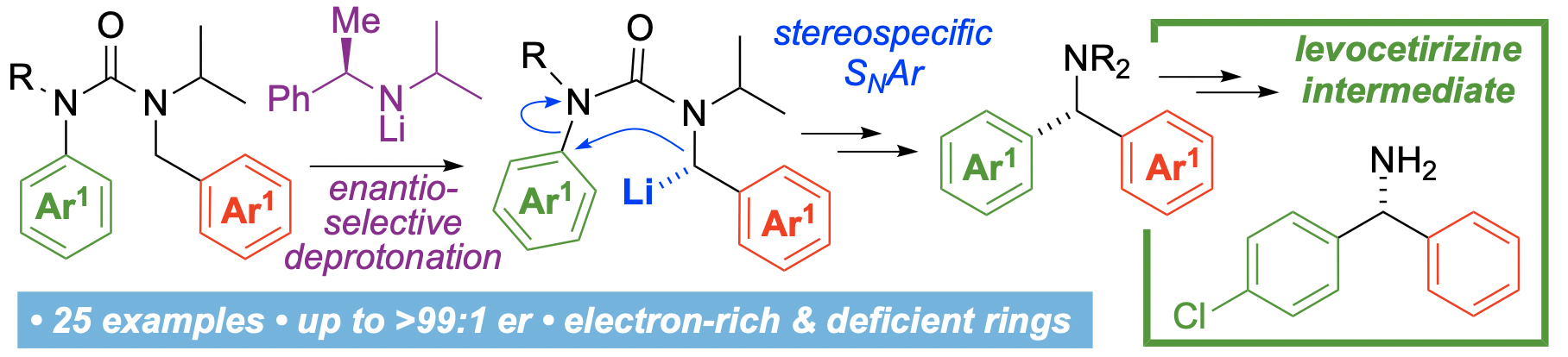
314. Enantioselective intramolecular α-arylation of benzylamine derivatives: synthesis of a precursor to levocetirizine
Rakesh K. Saunthwal, Maria Schwarz, Rajendra K. Mallick, William Terry-Wright, Jonathan Clayden
Angew. Chem. Int. Ed. 2023, 62, e202216758 [10.1002/anie.202216758]
2022
313. Dynamic Kinetic Resolution and Dynamic Kinetic Asymmetric Transformation of Atropisomers
Jordan Berreur, Beatrice S. L. Collins and Jonathan Clayden
in Science of Synthesis: Dynamic Kinetic Resolution (DKR) and Dynamic Kinetic Asymmetric Transformations (DYKAT), Bäckvall, J.-E., Ed.; Thieme: Stuttgart (2023); Vol. 1, 441–483 [10.1055/sos-SD-237-00162]
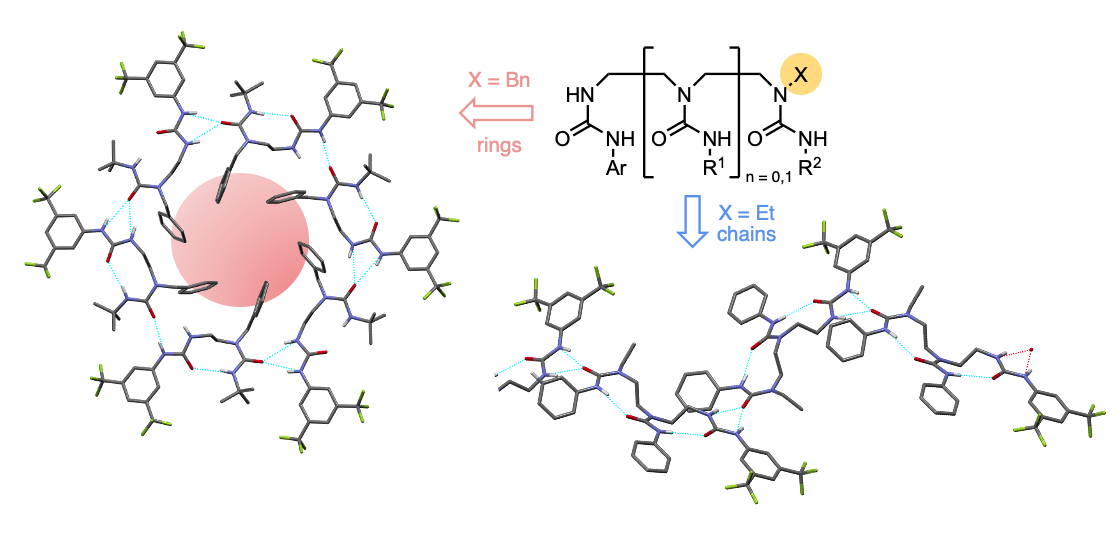
312. Supramolecular interactions between ethylene-bridged oligoureas: nanorings and chains formed by cooperative positive allostery
David P. Tilly, Matej Žabka, Inigo Vitorica-Yrezabal, Hazel A. Sparkes, Natalie Pridmore and Jonathan Clayden
Chem. Sci. 2022, 13, 13153-13159 [10.1039/D2SC04716K]
311. Biocatalytic Enantioselective Synthesis of Atropisomers
Acc. Chem. Res. 2022, 55, 3362–3375 [10.1021/acs.accounts.2c00572]

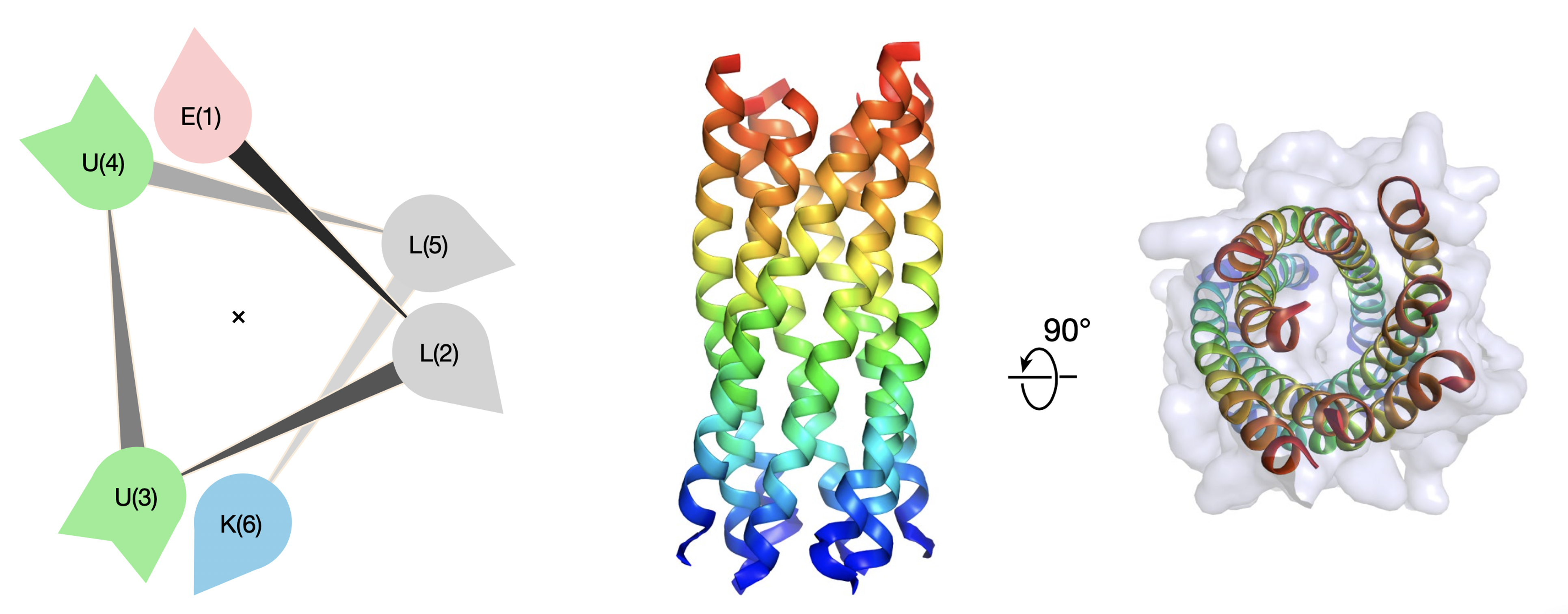
310. De novo design of discrete, stable 310-helix peptide assemblies
Prasun Kumar, Neil G. Paterson, Jonathan Clayden and Derek N. Woolfson
Nature 2022, 607, 387-392 [10.1038/s41586-022-04868-x]
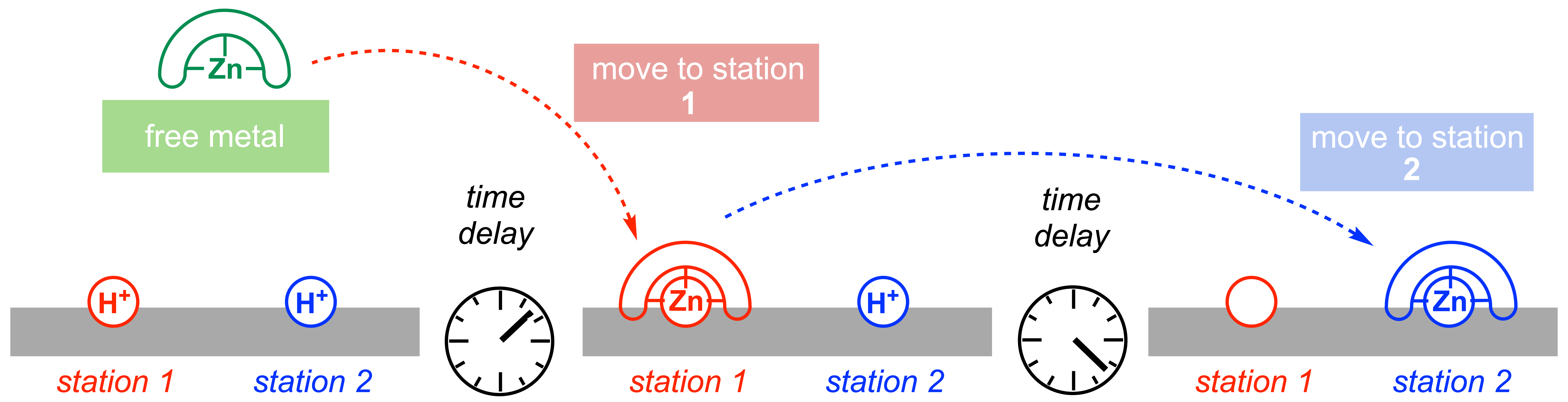
309. A Chemically Fuelled Molecular Automaton Displaying Programmed Migration of Zn2+ Between Alternative Binding Sites
Matthew M. Wootten, Sofja Tshepelevitsh, Ivo Leito, and Jonathan Clayden
Chem. Eur. J. 2022, 28, e202203347 [doi 10.1002/chem.202202247]
308. Hydrogen Bond Chains in Foldamers and Dynamic Foldamers
David T. J. Morris and Jonathan Clayden
in Spectroscopy and Computation of Hydrogen-Bonded Systems, ed M. J. Wojcik and Y. Ozaki, Wiley-VCH, 2022, p. 479-520: ISBN: 978-3-527-34972-2
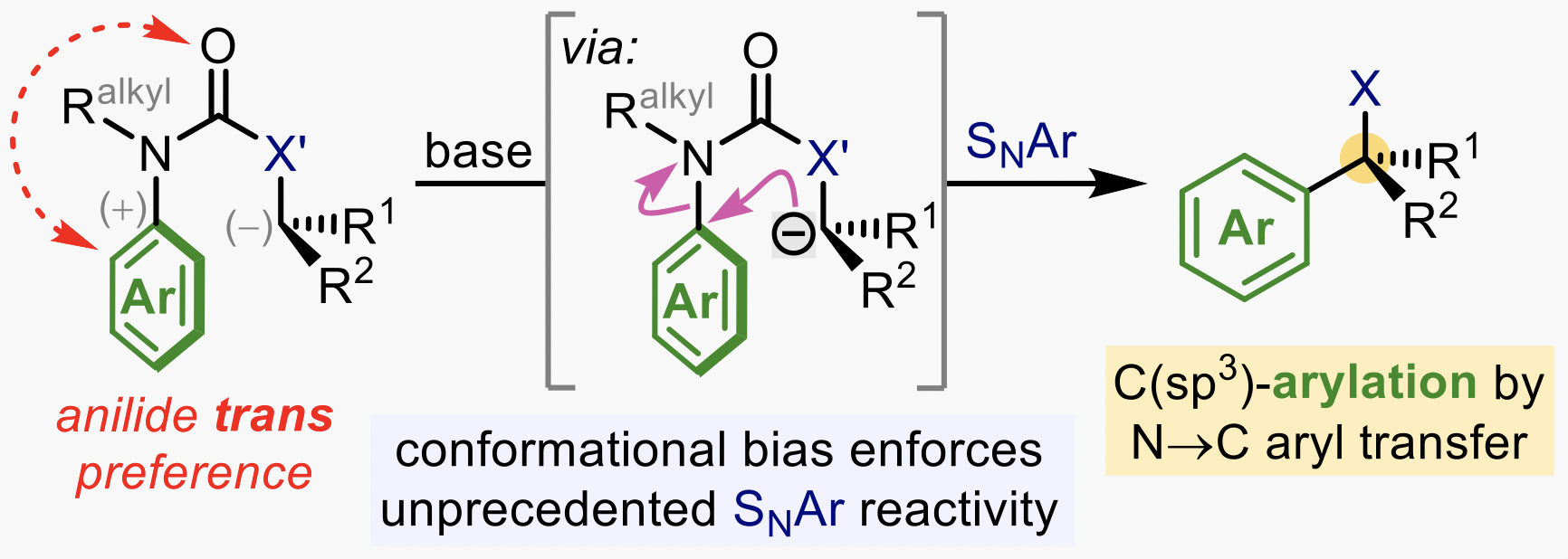
307. C(sp3)-Arylation by Conformationally Accelerated Intramolecular Nucleophilic Aromatic Substitution (SNAr)
Steven M. Wales, Rakesh K. Saunthwal and Jonathan Clayden
Acc. Chem. Res. 2022, 55, 1731–1747 [doi 10.1021/acs.accounts.2c00184]
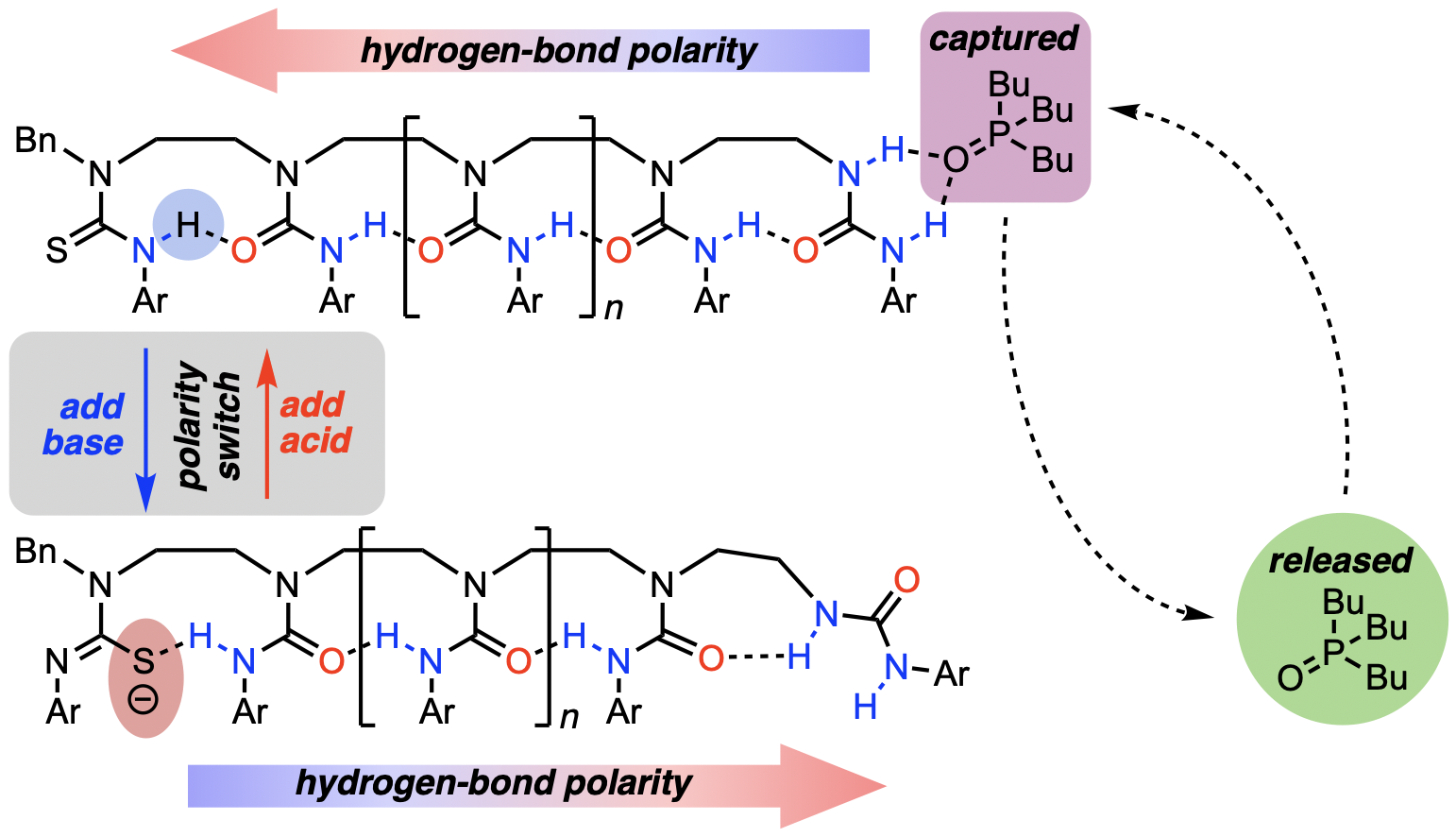
306. Reversible capture and release of a ligand mediated by a long-range relayed polarity switch in a urea oligomer
Steven M. Wales, David T. J. Morris and Jonathan Clayden
J. Am. Chem. Soc. 2022, 144, 2841–2846 [doi 10.1021/jacs.1c11928]
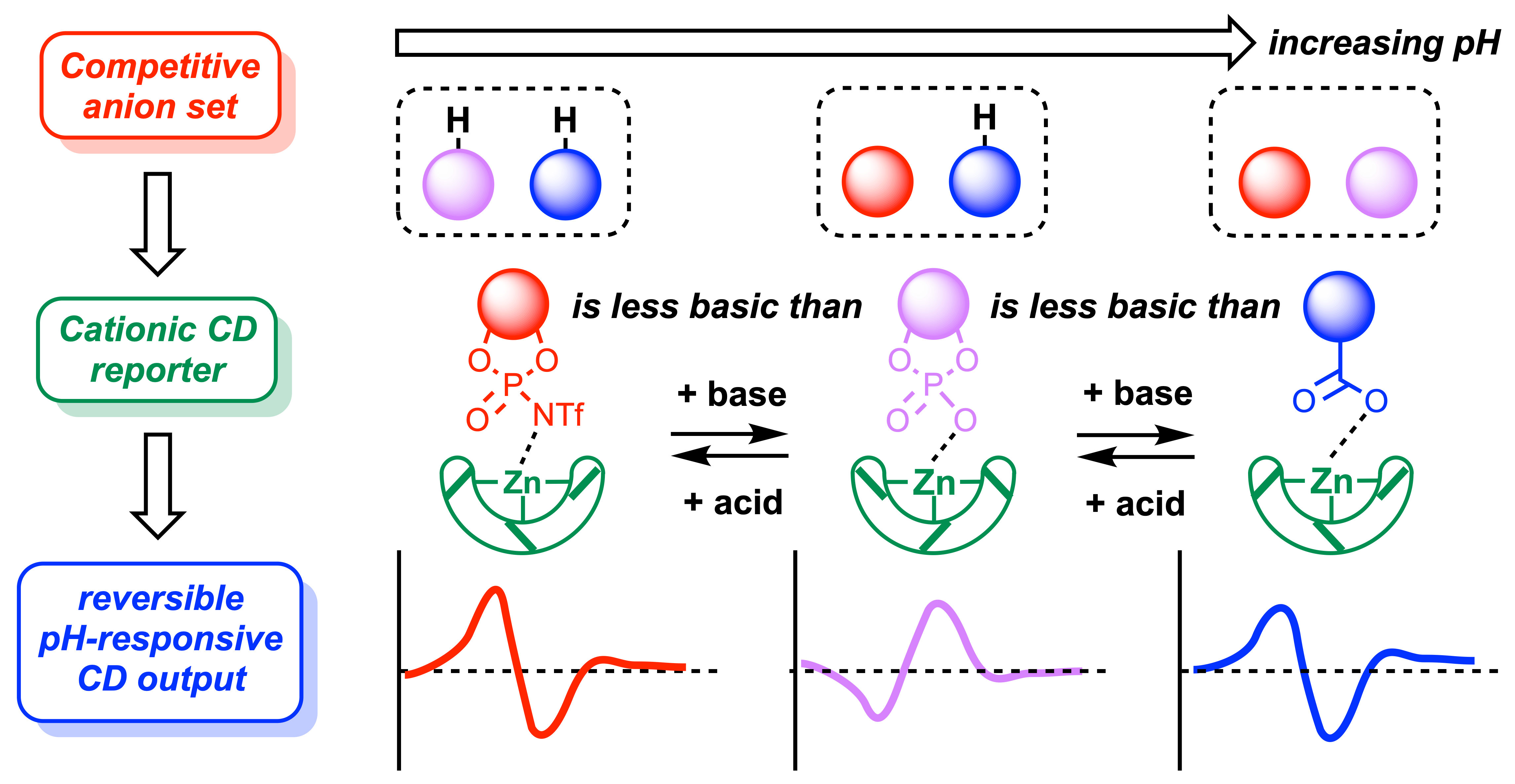
305. Inducing a pH-dependent conformational response by competitive binding to Zn2+ of a series of chiral ligands of disparate basicity
Matthew M. Wootten, Bryden A. F. Le Bailly, Sofja Tshepelevitsh, Ivo Leito and Jonathan Clayden
Chem. Sci. 2022, 13, 2258 – 2269[doi 10.1039/D1SC06812A]
[‘Pick of the week’ 9 Feb 2022]
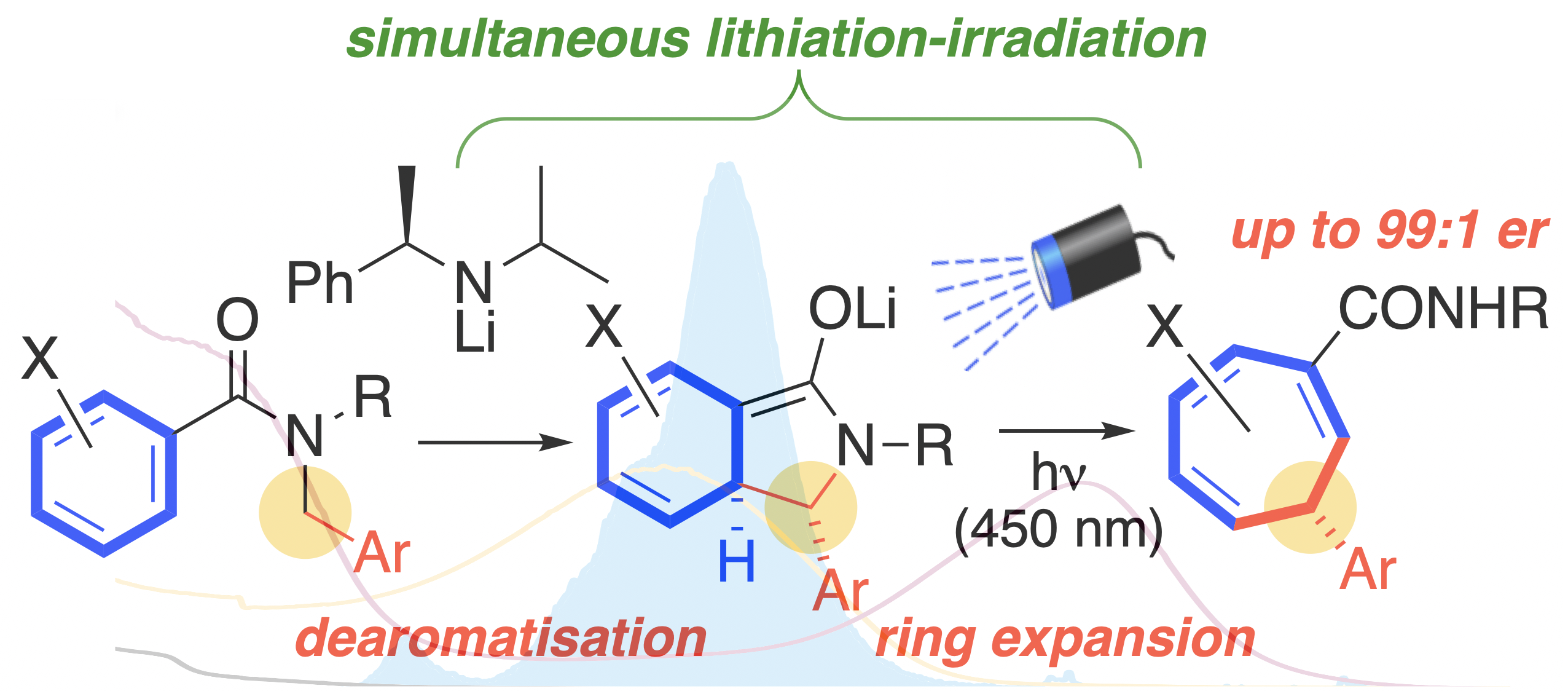
304. Enantioselective one-carbon expansion of aromatic rings by simultaneous formation and chromoselective irradiation of a transient coloured enolate
Rakesh K. Saunthwal, James Mortimer, Andrew J. Orr-Ewing and Jonathan Clayden
Chem. Sci. 2022, 13, 2079-2085 [doi 10.1039/D1SC06684F]
2021
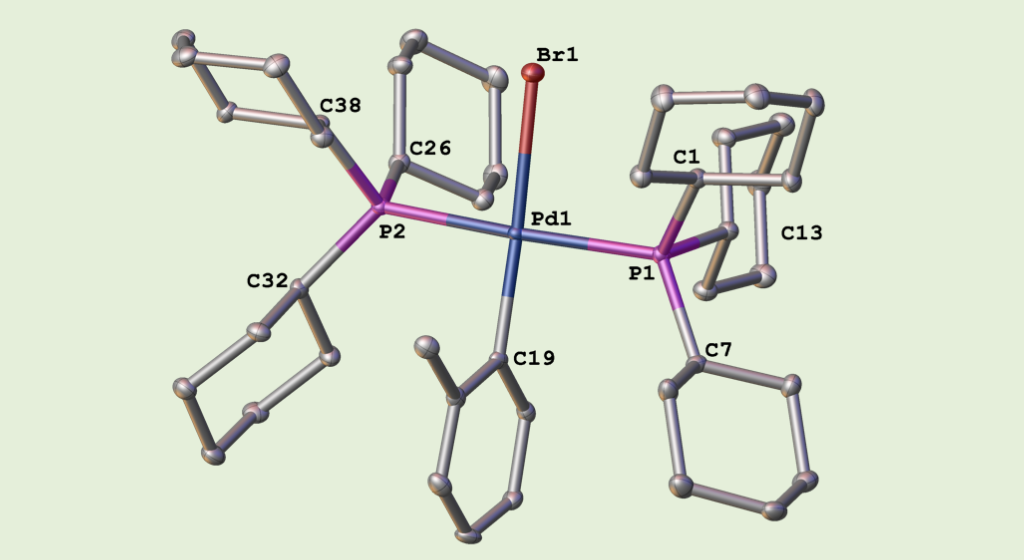
303. Identifying palladium culprits in amine catalysis
Mickaël Avanthay, Robin B. Bedford, Callum S. Begg, Dietrich Böse, Jonathan Clayden, Sean A. Davis, Jean-Charles Eloi, Georgy P. Goryunov, Ingo V. Hartung, Joseph Heeley, Kirill A. Khaikin, Matthew O. Kitching, Johannes Krieger, Pavel S. Kulyabin, Alastair J. J. Lennox, Roberto Nolla-Saltiel, Natalie E. Pridmore, Benjamin J. S. Rowsell, Hazel A. Sparkes, Dmitry V. Uborsky, Alexander Z. Voskoboynikov, Mark P. Walsh and Harry J. Wilkinson
Nature Catal. 2021, 4, 994-998 [doi 10.1038/s41929-021-00710-1]
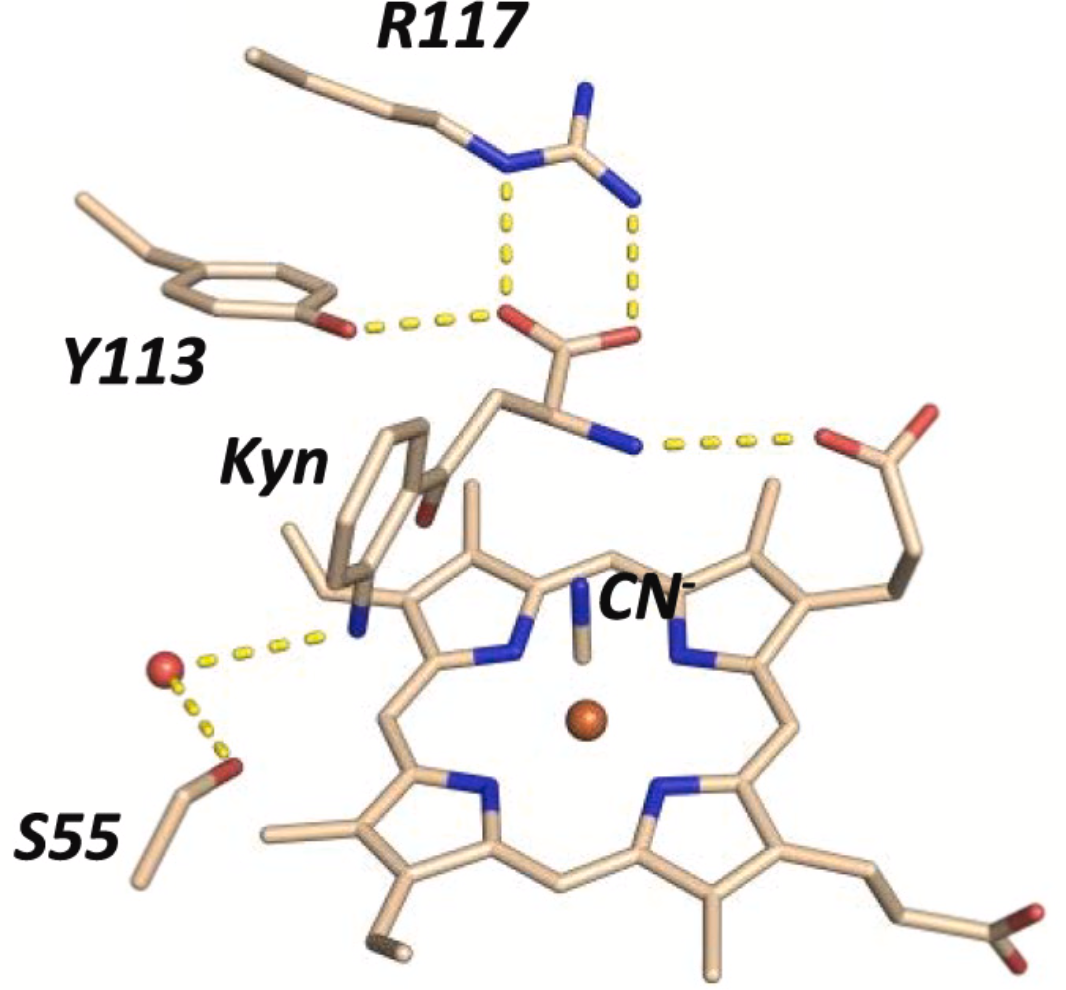
302. Binding of L-kynurenine to X. campestris tryptophan 2,3-dioxygenase
Jaswir Basran, Elizabeth S. Booth, Laura P. Campbell. Sarah J. Thackray, Mehul H. Jesani, Jonathan Clayden, Peter C. E. Moody, Christopher G. Mowat, Hanna Kwon, and Emma L. Raven
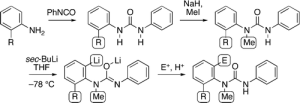
301. ‘Reverse biomimetic’ synthesis of L-arogenate and its stabilized analogues from L-Tyrosine
Louise Eagling, Daniel J. Leonard, Maria Schwarz, Iñaki Urruzuno, Grace Boden, J. Steven Wailes, John W. Ward, and Jonathan Clayden
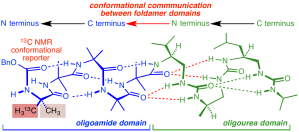
300. A Molecular Communication Channel Consisting of a Single Reversible Chain of Hydrogen Bonds in a Conformationally Flexible Oligomer
David T. J. Morris, Steven M. Wales, David Tilly, Elliot H. E. Farrar, Matthew N. Grayson, John W. Ward, and Jonathan Clayden
Chem 2021, 7, 2460-2472 [doi 10.1016/j.chempr.2021.06.022]
[Accompanied by ‘Highlight’ article: C. Nieuwland, C. F. Guerra Chem 2021, 7, 2272-2274 doi 10.1016/j.chempr.2021.07.015]
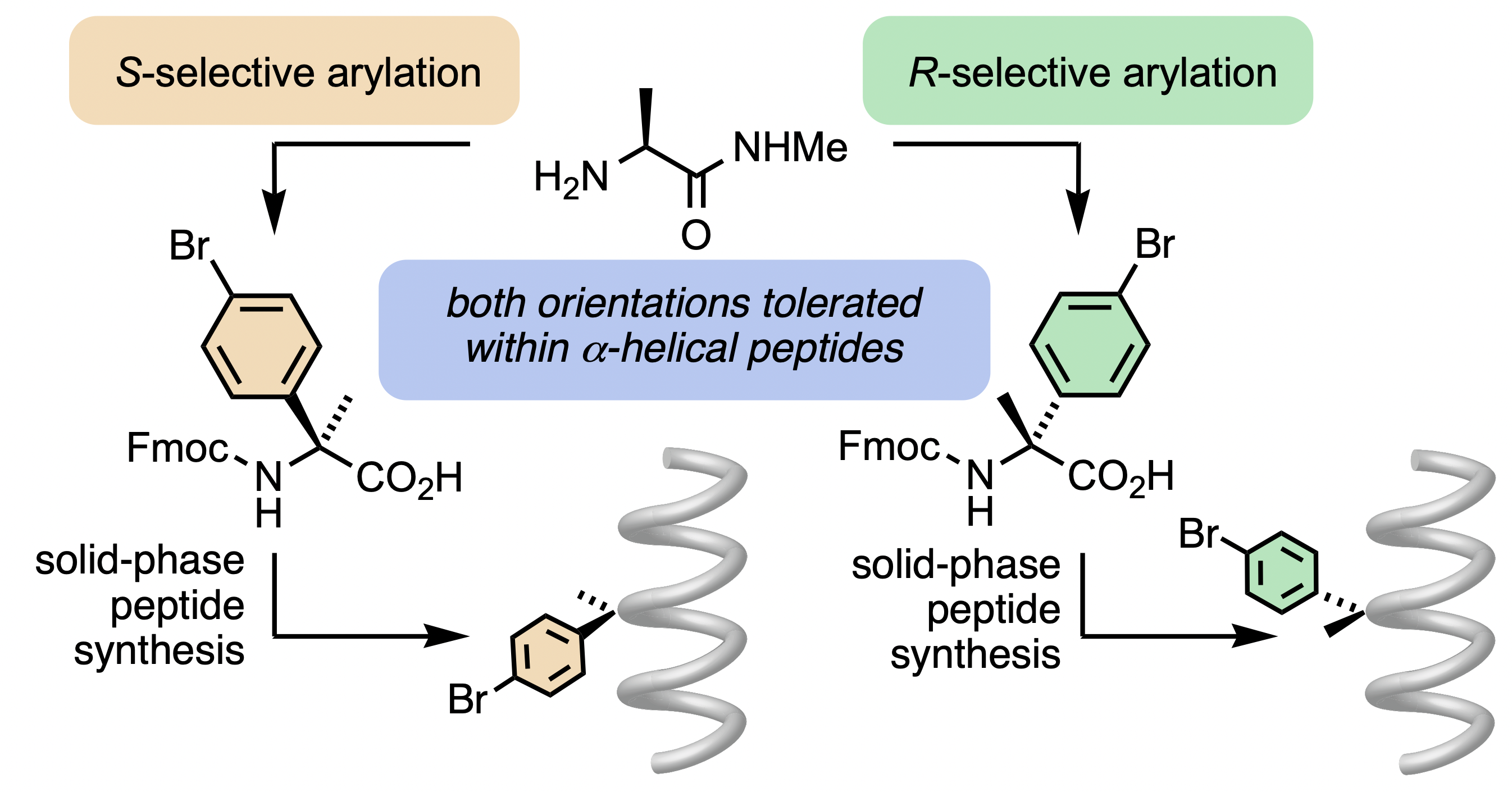
299. Scalable synthesis and coupling of quaternary alpha-arylated amino acids (Q4As): alpha-aryl substituents are tolerated in alpha-helical peptides
Daniel J. Leonard, Francis Zieleniewski, Isabelle Wellhöfer, Emily G. Baker, John W. Ward, Derek N. Woolfson, and Jonathan Clayden
Chem. Sci., 2021, 12, 9386-9390 [doi 10.1039/D1SC01378E]
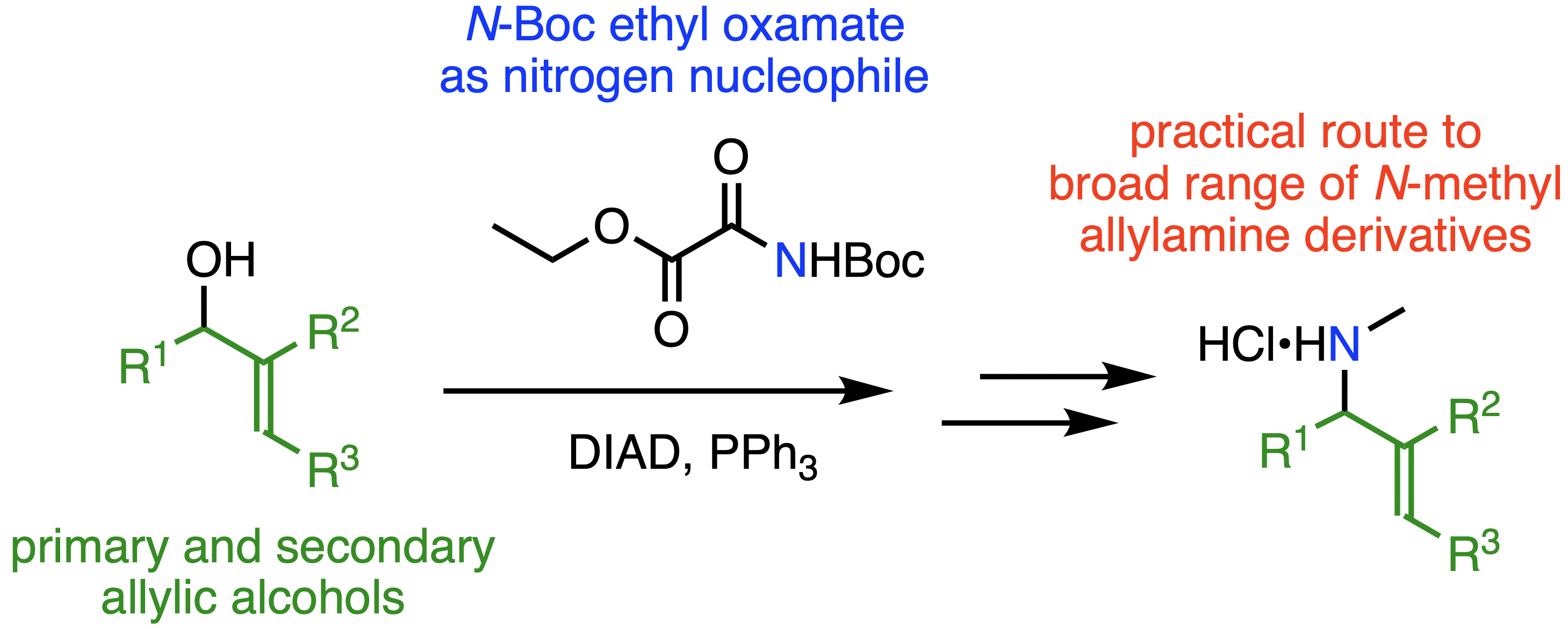
298. N-Methyl Allylic Amines from Allylic Alcohols by Mitsunobu Substitution using N-Boc Ethyl Oxamate
Branca C. van Veen, Steven M. Wales, and Jonathan Clayden
J. Org. Chem. 2021, 86, 8538-8543 [doi 10.1021/acs.joc.1c00918]
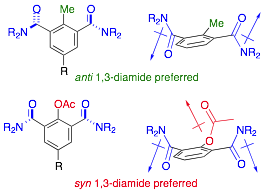
297. Triarylmethanes and their Medium-Ring Analogues by Unactivated Truce-Smiles Rearrangement of Benzanilides
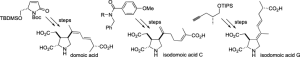
Roman Abrams, Mehul H. Jesani, Alex Browning, and Jonathan Clayden
Angew. Chem. Int. Ed., 2021, 60, 11272-11277 [doi 10.1002/anie.202102192]

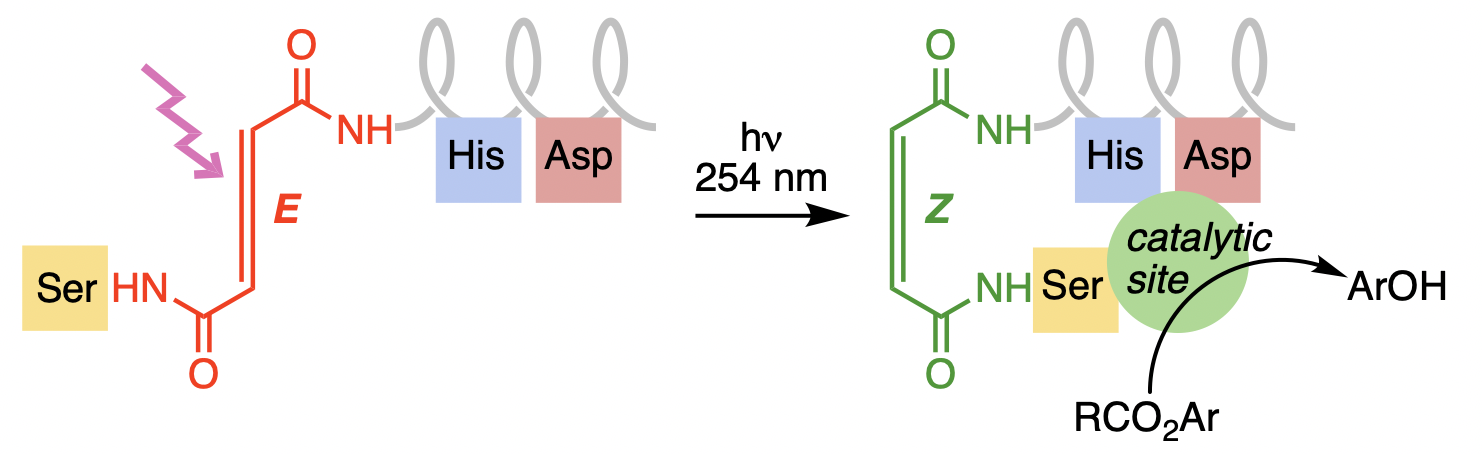
296. Light-mediated control of activity in a photosensitive foldamer that mimics an esterase
Matteo Pollastrini, Giulia Marafon, Jonathan Clayden, and Alessandro Moretto
Chem. Commun. 2021, 57, 2269-2272 [doi 10.1039/d0cc08309g]
295. Insight into mechanism of action and peptide-membrane interactions of Aib-rich peptides: multi-technique experimental and theoretical analysis
Maria Giovanna Lizio, Mario Campana, Matteo De Poli, Damien F. Jefferies, William Cullen, Valery Andrushchenko, Nikola P. Chmel, Petr Bouř, Syma Khalid, Jonathan Clayden, Ewan Blanch, Alison Rodger, and Simon J. Webb
294. Hydantoin-bridged medium ring scaffolds by migratory insertion of urea-tethered nitrile anions into aromatic C-N bonds
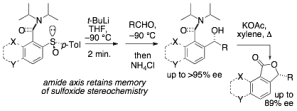
Makenzie J. Millward, Emily Ellis, John W. Ward and Jonathan Clayden
Chem. Sci. 2021, 12, 2091-2096 [doi 10.1039/D0SC06188C]

2020
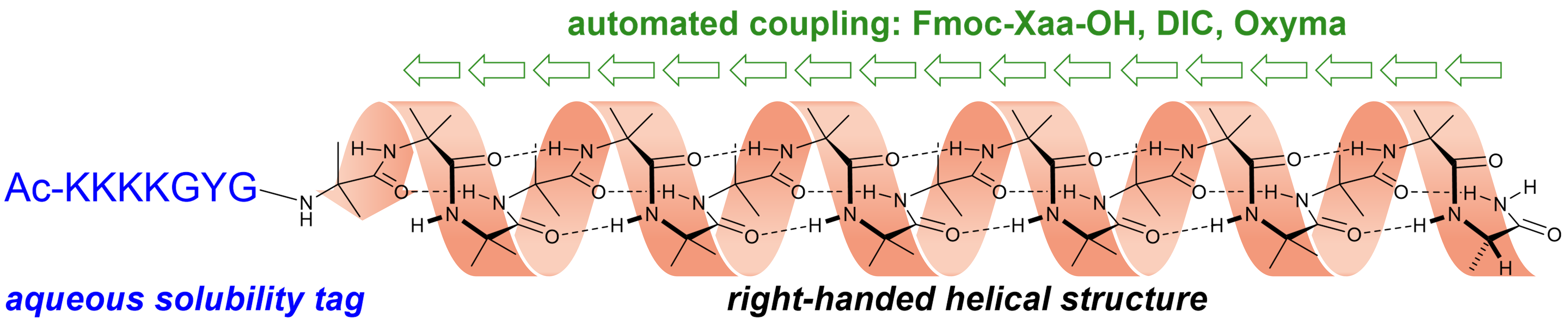
293. Automated solid-phase concatenation of Aib residues to form long, water-soluble, helical peptides
Francis Zieleniewski, Derek N. Woolfson and Jonathan Clayden
Chem. Commun. 2020, 56, 12049-12052 [doi 10.1039/D0CC04698A]
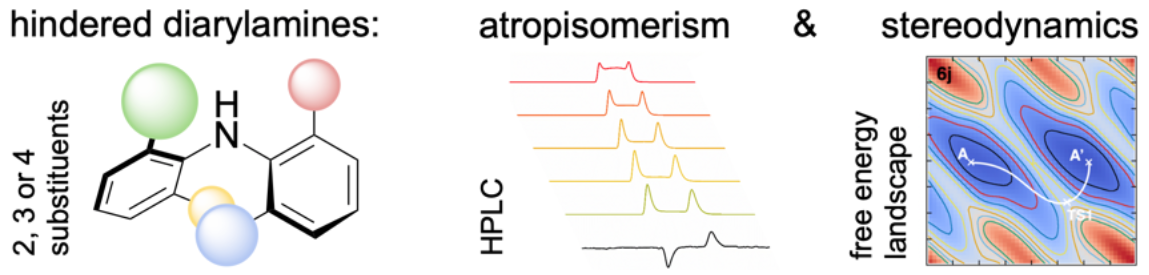
292. Atropisomerism in diarylamines: structural requirements and mechanisms of conformational interconversion
Romain Costil, Alistair Stirling, Fernanda Duarte and Jonathan Clayden
Angew. Chemie. Int. Ed. 2020, 59, 18670-18678 [doi 10.1002/anie.202007595]
291. Switchable foldamer ion channels with anion selectivity and antibiotic activity
Anna D. Peters, Stefan Borsley, Flavio della Sala, Dominic Cairns-Gibson, Marios Leonidou, Jonathan Clayden, George F. S. Whitehead, Iñigo J. Vitorica-Yrezabal, Eriko Takano, John Burthem, Scott L. Cockroft, and Simon J. Webb
Chem. Sci. 2020, 11, 7023-7030 [doi 10.1039/D0SC02393K]
290. Photocatalytic Difunctionalization of Vinyl Ureas by Radical Addition Polar Truce-Smiles Rearrangement Cascades
Roman Abrams and Jonathan Clayden
Angew. Chemie. Int. Ed. 2020, 59, 11600-11606 [doi 10.1002/anie.202003632]
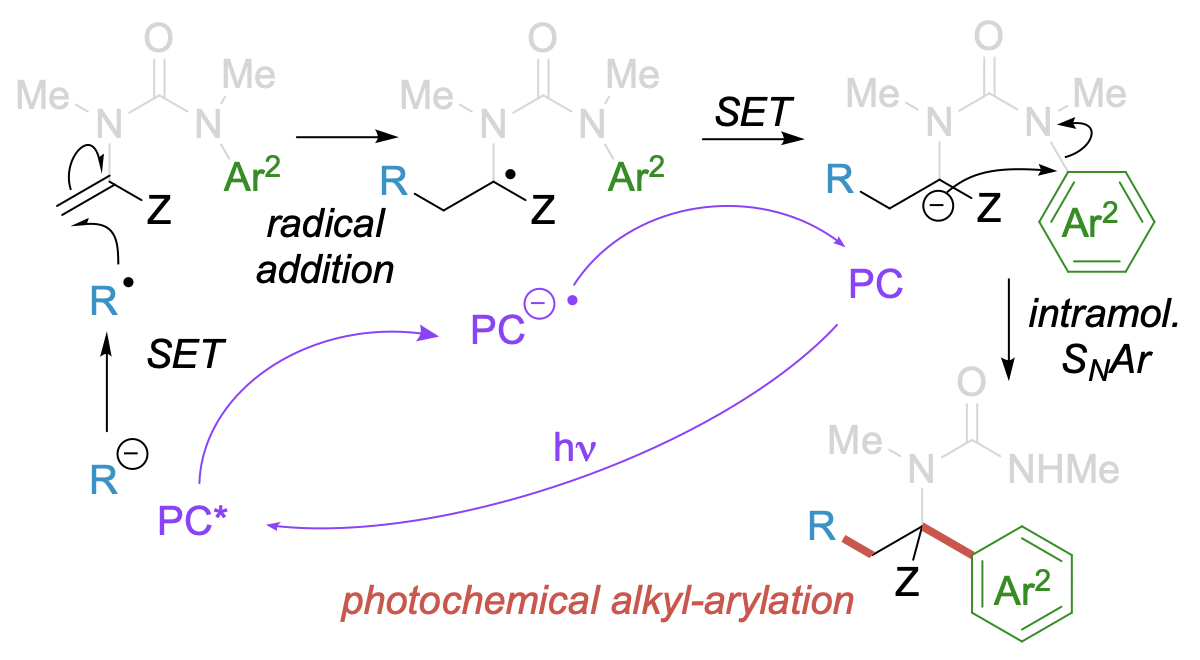
289. Molecular Recognition by Zn(II)-Capped Dynamic Foldamers
Natasha Eccles, Flavio della Sala, Bryden A. F. Le Bailly, George F. S. Whitehead, Jonathan Clayden and Simon J. Webb
Chem. Open 2020, 9, 338 [doi 10.1002/open.201900362]
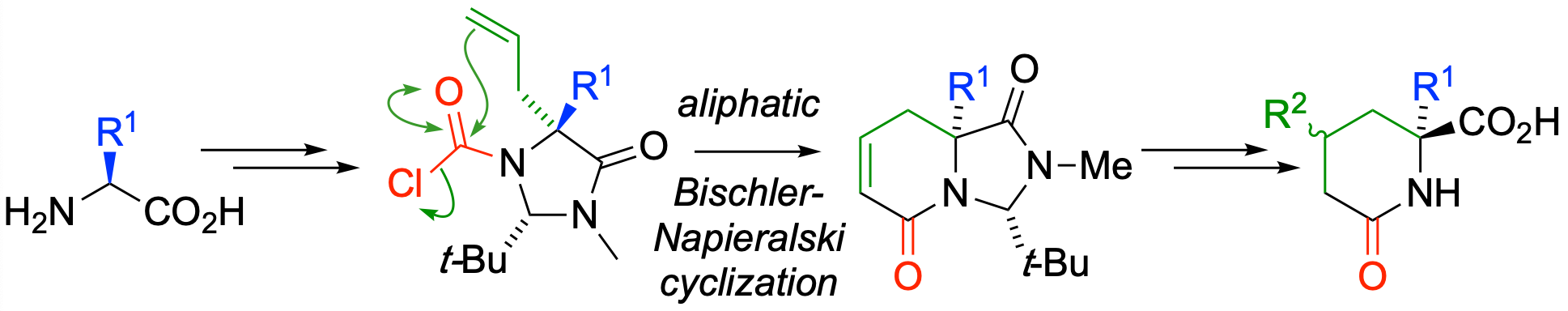
288. An aliphatic Bischler-Napieralski reaction: dihydropyridones by cyclocarbonylation of 3-allylimidazolidin-4-ones
Mostafa M. Amer, Olatz Olaizola, Jennifer Carter, Hossay Abas and Jonathan Clayden
Org. Lett. 2020, 22, 253-256 [doi 10.1021/acs.orglett.9b04250]
287. Stuart Warren: 24 Dec 1938 – 22 Mar 2020
Varinder K. Aggarwal, Susan K. Armstrong, Lorenzo Caggiano, Kelly Chibale, Jonathan Clayden, Iain Coldham, Nicholas Greeves, Richard C. Hartley, Julian G. Knight, Nikolai Kuhnert, Helen J. Mitchell, Adam Nelson, Peter O’Brien, Stephen P. Thomas and Paul Wyatt

2019
286. Fluorine and amide groups together at last
Jonathan Clayden
Nature 2019, 573, 37-38 [News and Views article; doi 10.1038/d41586-019-02611-7]
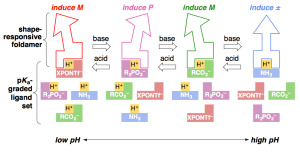
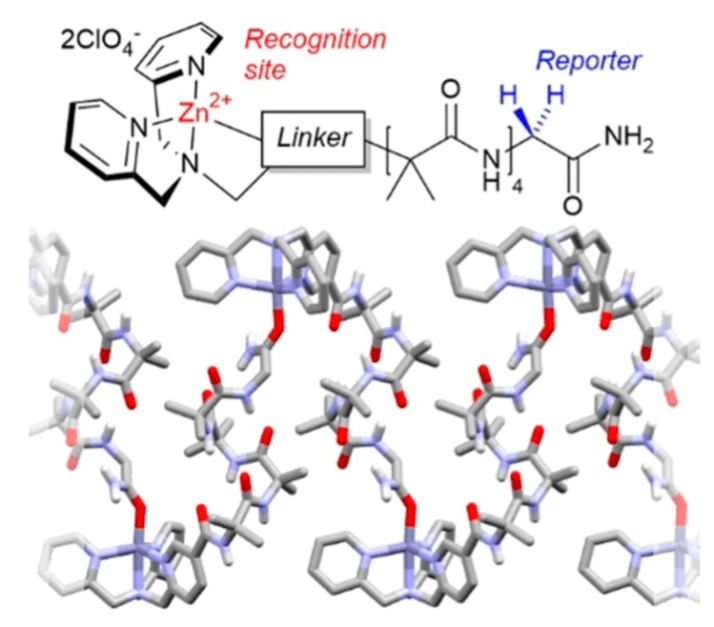
285. Remote conformational responses to enantiomeric excess in carboxylate-binding dynamic foldamers
Natasha Eccles, Bryden A. F. Le Bailly, Flavio della Sala, Iñigo J. Vitórica-Yrezábal, Jonathan Clayden and Simon J. Webb
Chem. Commun. 2019, 55, 9331-9334 [doi 10.1039/C9CC03895G]
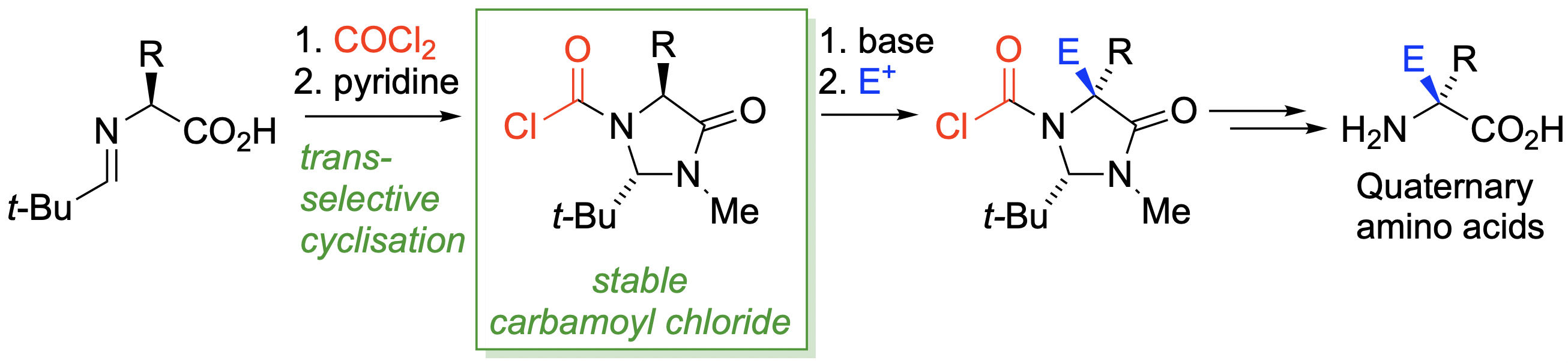
284. Amino acid-derived trans–N-chloroformylimidazolidinones: scalable, stereoselective synthesis, structure, and utility
Mostafa Mahmoud Amer, Hossay Abas, Daniel J. Leonard, John W. Ward and Jonathan Clayden
J. Org. Chem. 2019, 84, 117199-7206 [doi 10.1021/acs.joc.9b00727]
283. Extended diethylglycine homopeptides formed by desulfurization of their tetrahydrothiopyran analogues
Marta De Zotti and Jonathan Clayden
Org. Lett. 2019, 21, 2209-2212 [doi 10.1021/acs.orglett.9b00501]
282. N-Chloroformylimidazolidinone Enolates as 1,3-Dipolar Reagents for the Stereoselective Synthesis of 3,4-Dihydroisoquinolones
Hossay Abas, Mostafa Mahmoud Amer, Olatz Olaizola and Jonathan Clayden
Org. Lett. 2019, 21, 1908-1911 [doi 10.1021/acs.orglett.9b00548]
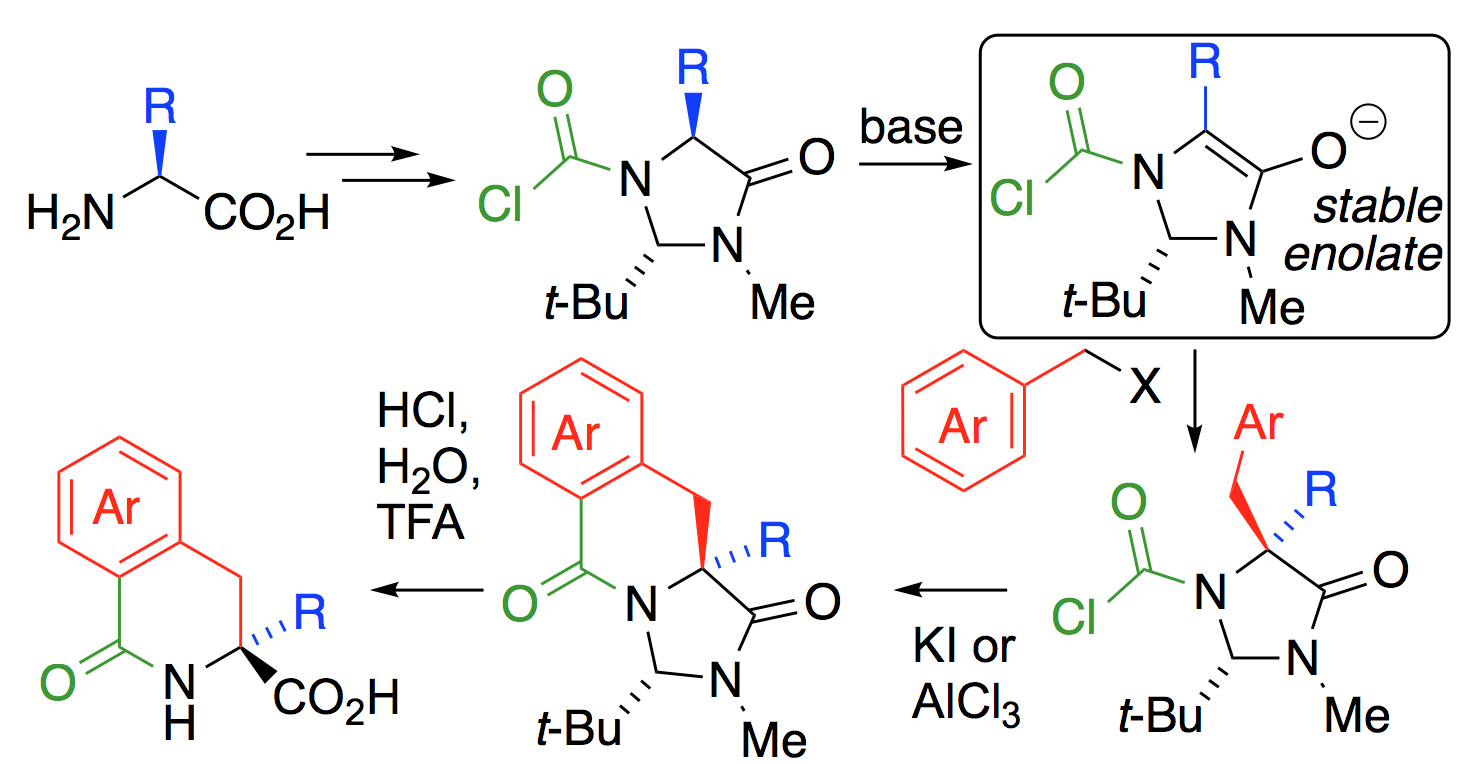
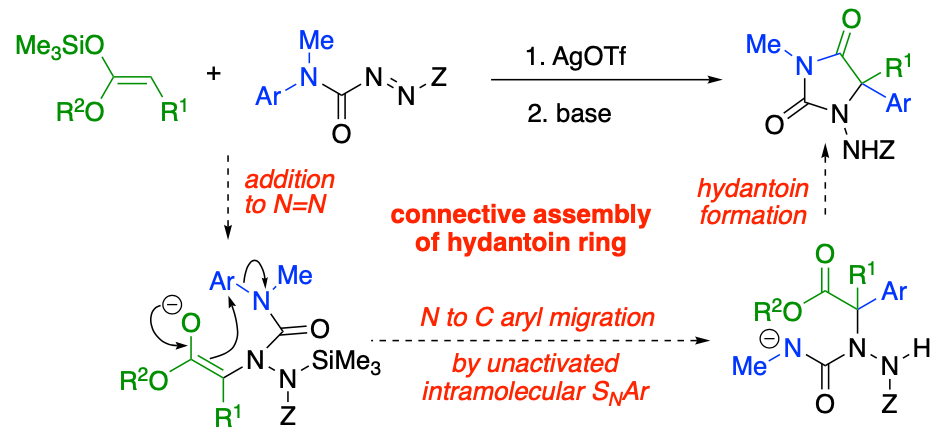
281. Connective Synthesis of 5,5-Disubstituted Hydantoins by Tandem α-Amination and α-Arylation of Silyl Ketene Acetals
Rakesh K. Saunthwal, Matthew T. Cornall, Roman Abrams, John W. Ward and Jonathan Clayden
Chem. Sci. 2019, 10, 3408-3412 [doi 10.1039/C8SC05263H]
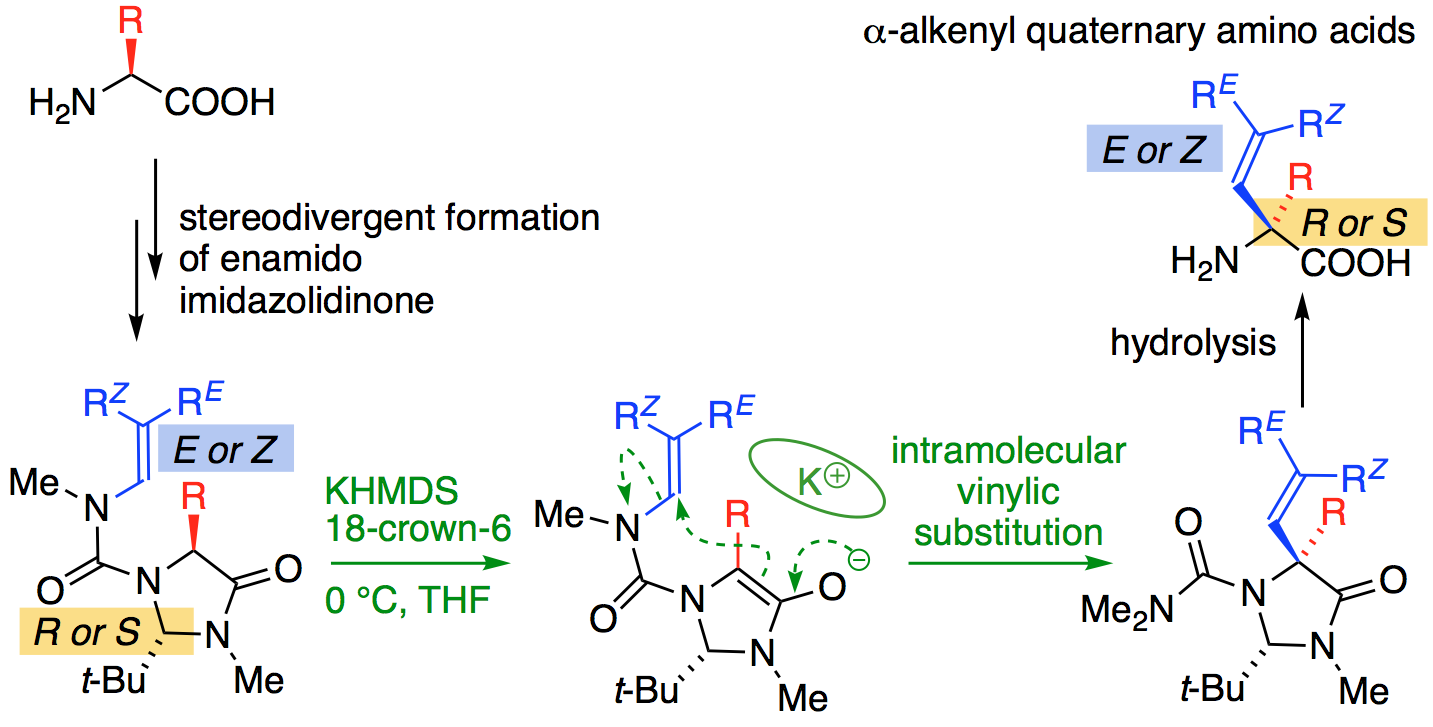
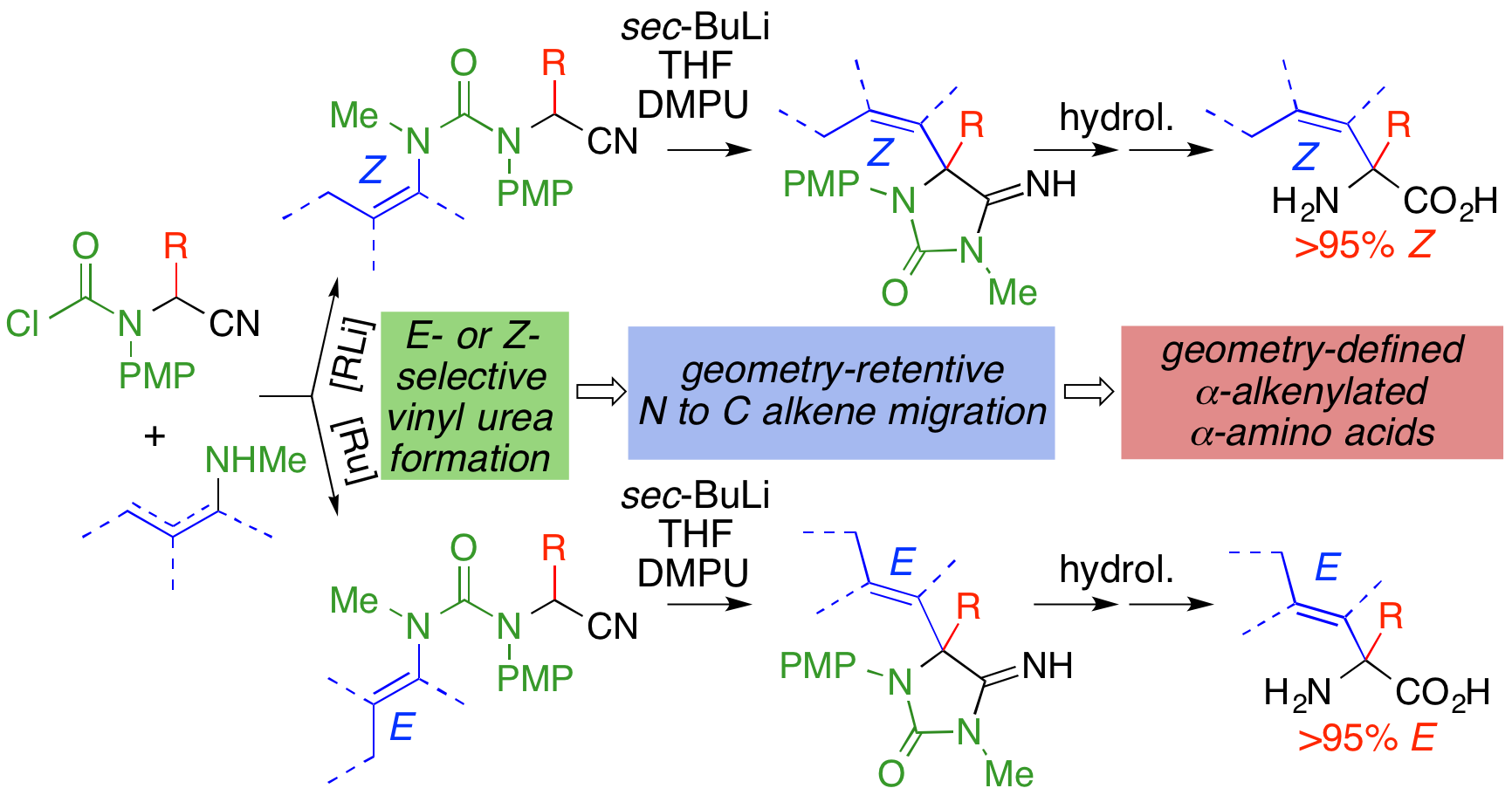
280. Asymmetric and Geometry-selective α-Alkenylation of α-Amino Acids
Hossay Abas, Josep Mas-Roselló, Mostafa M. Amer, Derek J. Durand, Robin R. Groleau, Natalie Fey, Jonathan Clayden
Angew. Chem. Int. Ed. 2019, 58, 2418-2422 [doi 10.1002/anie.201813984]
2018
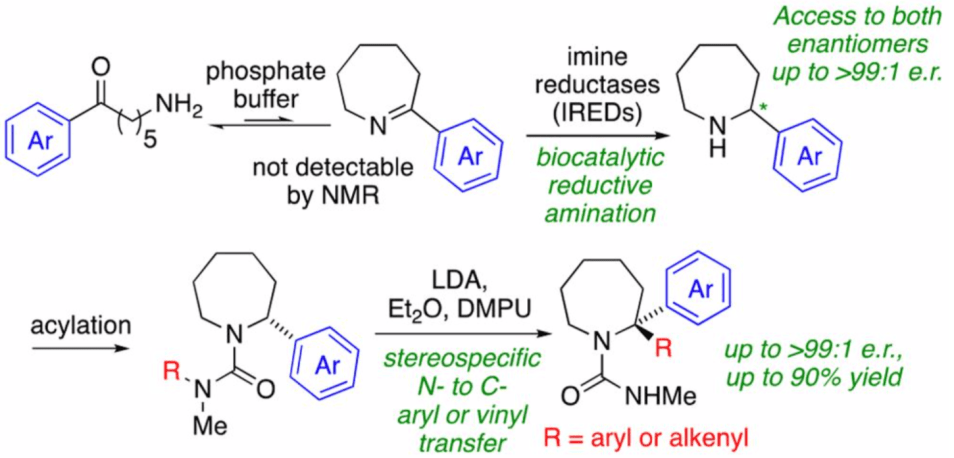
279. Chemoenzymatic synthesis of substituted azepanes by sequential biocatalytic reduction and organolithium-mediated rearrangement
Wojciech Zawodny, Sarah L. Montgomery, James R. Marshall, James D. Finnigan, Nicholas J. Turner and Jonathan Clayden
J. Am. Chem. Soc. 2018, 140, 17872–17877 [doi 10.1021/jacs.8b11891]
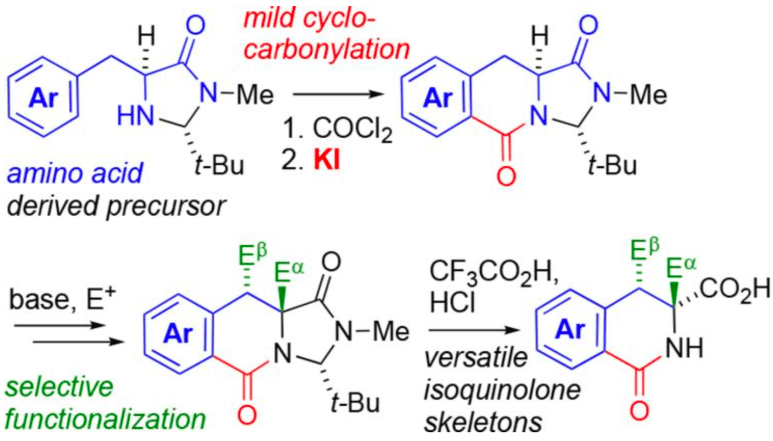
278. Substituted dihydroisoquinolinones by iodide-promoted cyclocarbonylation of aromatic alpha-amino acids
Mostafa M. Amer, Ana C. Carrasco, Daniel J. Leonard, John W. Ward and Jonathan Clayden
Org. Lett. 2018, 20, 7977-7981 [10.1021/acs.orglett.8b03551]
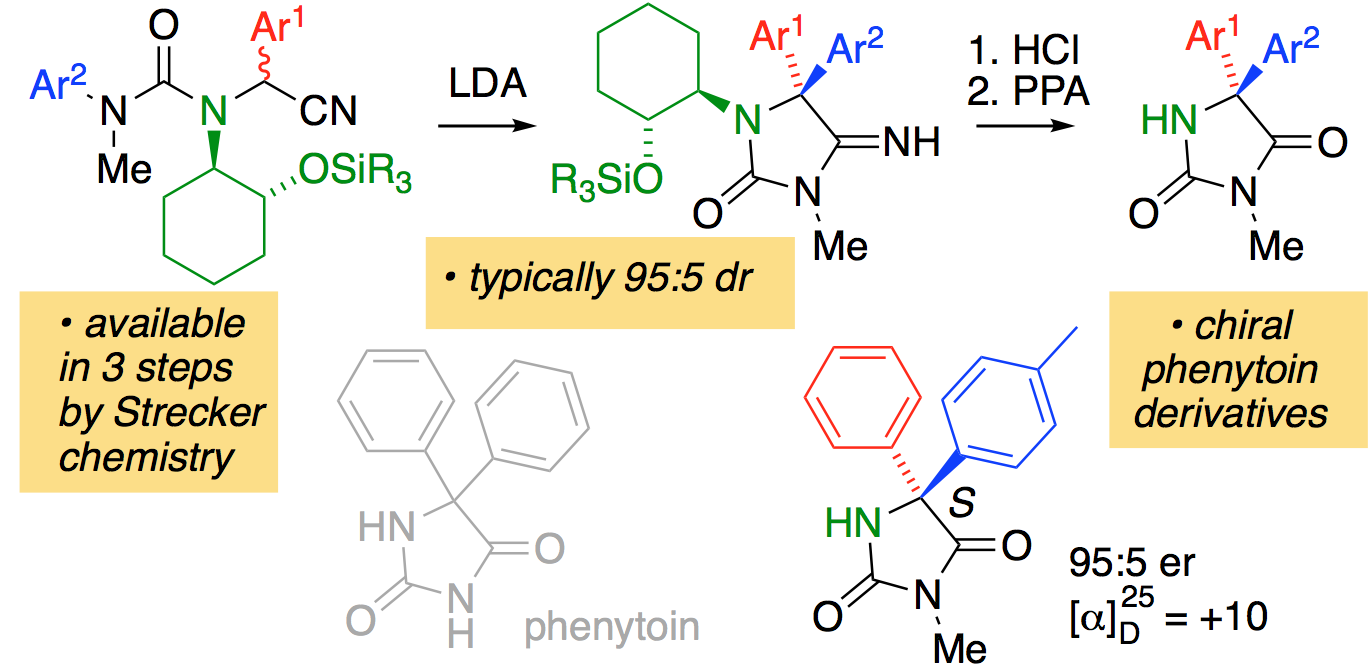
277. Enantioselectively functionalised phenytoin derivatives by diastereoselective intramolecular arylation of lithiated alpha-amino nitriles
Josep Mas-Roselló, Mary Okoh and Jonathan Clayden
Chem. Commun. 2018, 54, 10985-10988 [doi 10.1039/C8CC06833J]
Highlighted in Synfacts [doi 10.1055/s-0037-1609640]
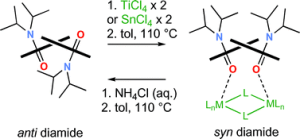
276. N,N’-Disuccinimidyl carbonate
Edwin C. Davison, Arun K. Ghosh, Nagaswamy Kumaragurubaran, David T. J. Morris, Jonathan Clayden
Encyclopedia of Reagents for Organic Synthesis 2018 [doi 10.1002/047084289X.rd469.pub3]
275. Asymmetric alpha-Arylation of Amino Acids
Daniel J. Leonard, John W. Ward and Jonathan Clayden
Nature 2018, 562, 105-109 [doi 10.1038/s41586-018-0553-9]
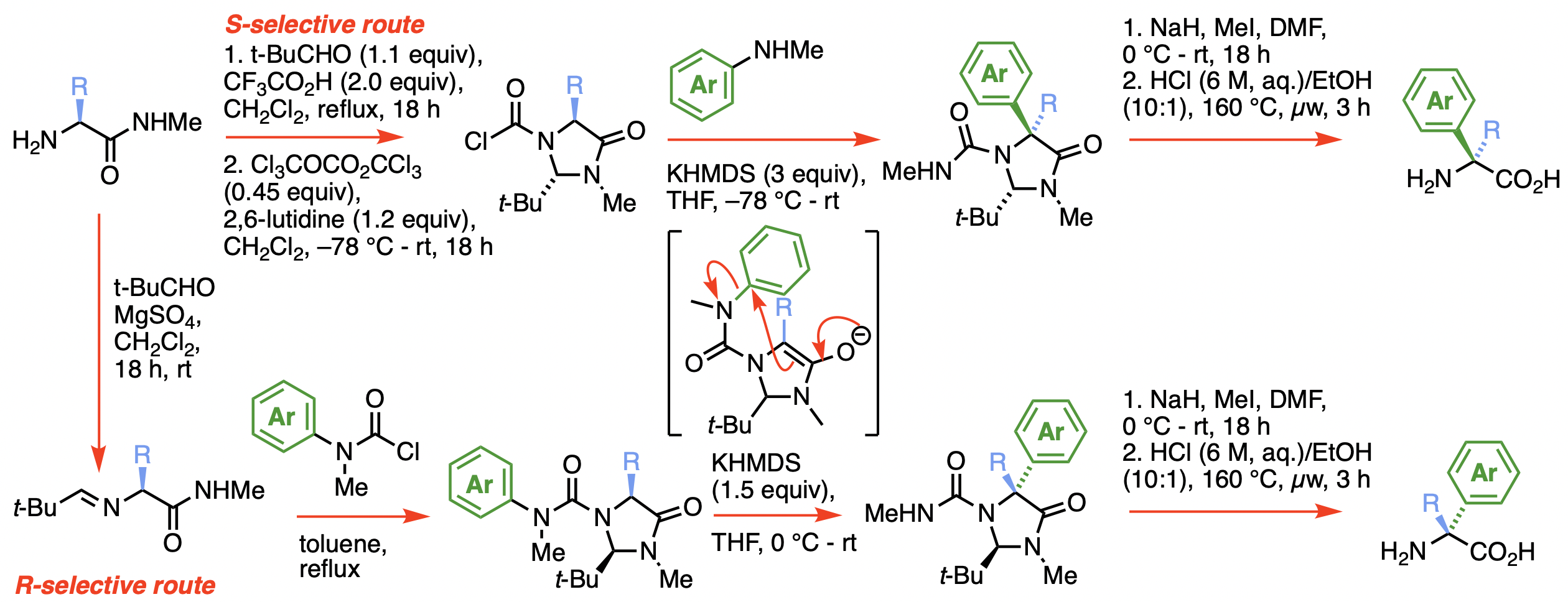
Highlighted in Synfacts 2019, 15, 0095 [doi 10.1055/s-0037-1611428]
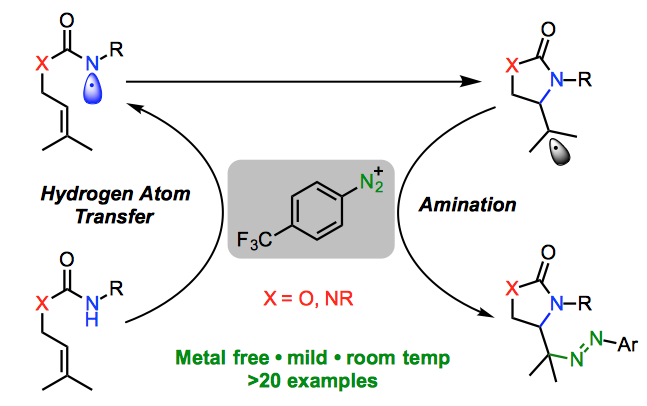
274. Transition Metal-Free Cycloamination of Prenyl Carbamates and Ureas Promoted by Aryldiazonium Salts
Roman Abrams, Quentin Lefebvre and Jonathan Clayden
Angew. Chemie Int. Ed. 2018, 57, 13587-13591 [doi 10.1002/anie.201809323]
273. Polycyclic indoline derivatives by dearomatizing anionic cyclization of indole and tryptamine-derived ureas
Jessica E. Hill, Quentin Lefebvre, Laura A. Fraser and Jonathan Clayden
Org. Lett. 2018, 20, 5770-5773 [doi 10.1021/acs.orglett.8b02468]

272. Bis-pyrene probes of foldamer conformation in solution and in phospholipid bilayers
Francis G. A. Lister, Natasha Eccles, Sarah J. Pike, Robert A. Brown, George F. S. Whitehead, James Raftery, Simon J. Webb and Jonathan Clayden
Chem. Sci. 2018, 9, 6860-6870 [doi 10.1039/C8SC02532K]
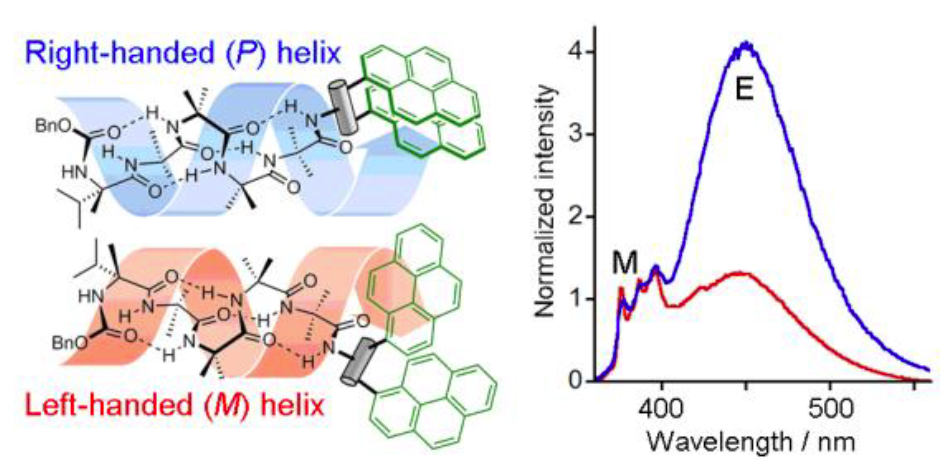
271. Optically Active Vibrational Spectroscopy of α-Aminoisobutyric Acid Foldamers in Organic Solvents and Phospholipid Bilayers
Maria Giovanna Lizio, Valery Andrushchenko, Sarah J. Pike, Anna D. Peters, George F. S. Whitehead, Iñigo J. Vitórica-Yrezábal, Shaun T. Mutter, Jonathan Clayden, Petr Bouř, Ewan W. Blanch, and Simon J. Webb
Chem. Eur. J. 2018, 24, 9399-9408. [doi 10.1002/chem.201801121]
270. Alpha-methyl phenylglycines by asymmetric alpha-arylation of alanine and their effect on the conformational preference of helical Aib foldamers
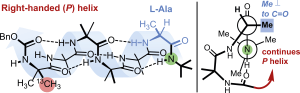
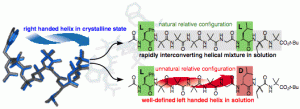
Romain Costil, Fernando Fernández-Nieto, Rachel C. Atkinson and Jonathan Clayden
Org. Biomol. Chem. 2018, 16, 2757-2761 [doi 10.1039/C8OB00551F]
269. Ring expansion and re-contraction for the synthesis of 1-aryl tetrahydroisoquinolines and tetrahydrobenzazepines from readily available heterocyclic precursors
Jessica E. Hill, Johnathan V. Matlock, Quentin Lefebvre, Katie G. Cooper and Jonathan Clayden
Angew. Chem. Int. Ed. 2018, 57, 5788-5791 [doi 10.1002/anie.201802188]
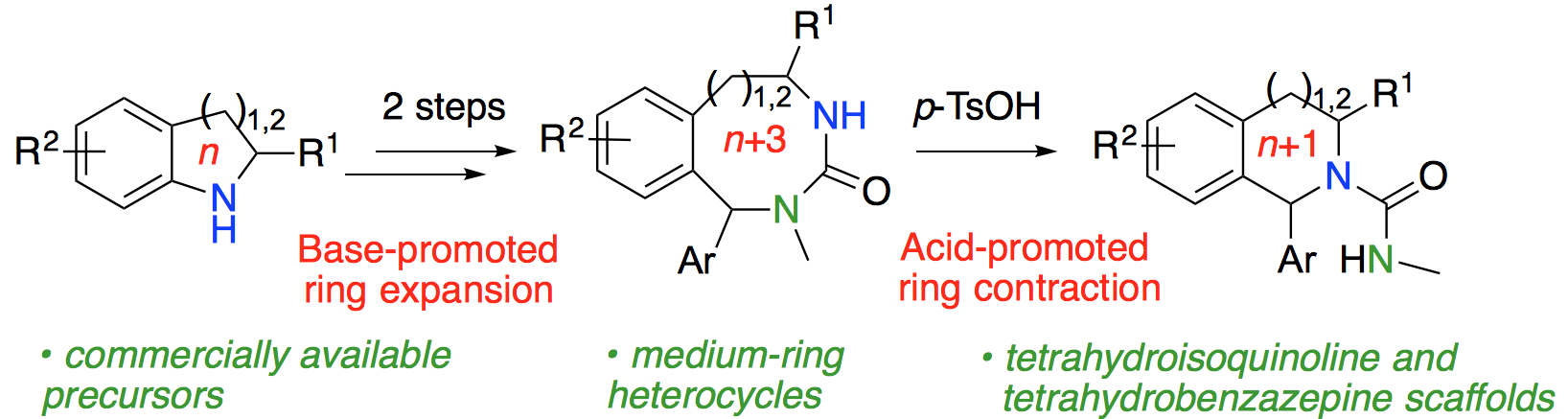
268. Competing hydrogen-bond polarities in a dynamic oligourea foldamer: a molecular spring torsion balance
Romina Wechsel, Matej Žabka, John W. Ward and Jonathan Clayden
J. Am. Chem. Soc. 2018, 140, 3528-3531 [doi 10.1021/jacs.8b00567]
267. The Role of Terminal Functionality in the Membrane and Antibacterial Activity of Peptaibol-Mimetic Aib Foldamers
Catherine Adam, Anna D. Peters, M. Giovanna Lizio, George F. S. Whitehead, James Cooper, Scott L. Cockroft, Jonathan Clayden and Simon J. Webb
Chem. Eur. J. 2018, 24, 2249-2256. [doi 10.1002/chem.201705299]
2017
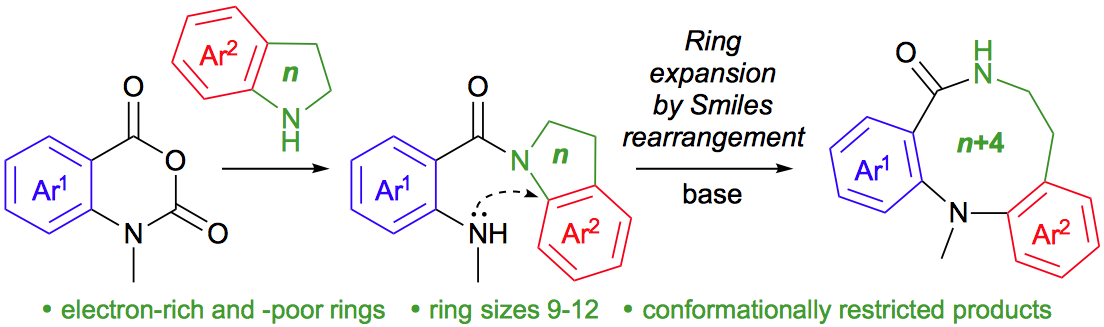
266. Medium-ring analogues of dibenzodiazepines by conformationally induced Smiles ring expansion
Romain Costil, Quentin Lefebvre, Jonathan Clayden
Angew. Chemie Int. Ed. 2017, 56, 14602–14606 [doi 10.1002/anie.201708991]
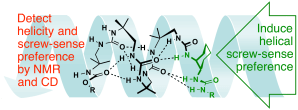
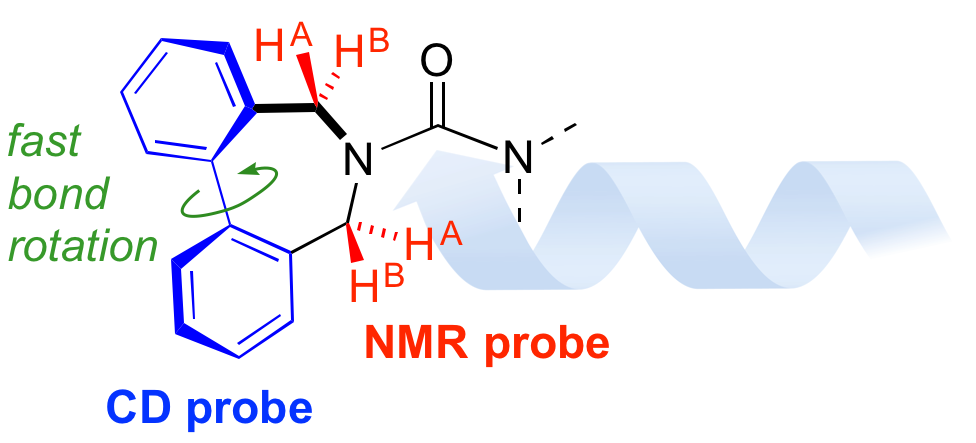
265. Dibenzazepinyl ureas as dual NMR and CD probes of helical screw-sense preference in conformationally equilibrating dynamic foldamers
Vincent Diemer, Julien Maury, Bryden A. F. Le Bailly, Simon J. Webb, Jonathan Clayden
Chem. Commun. 2017, 53, 10768-10771 [doi 10.1039/C7CC06427F] [Special Issue on Chemosensors and Molecular Logic]
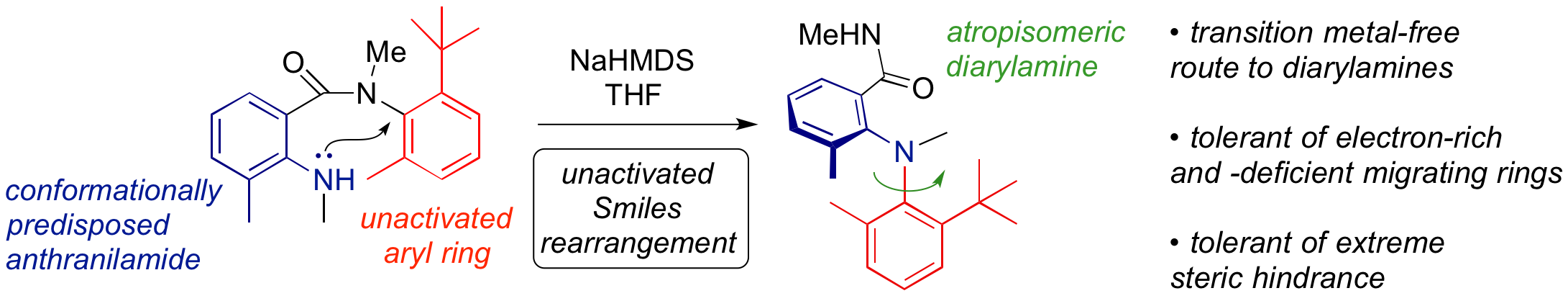
264. Heavily Substituted Atropisomeric Diarylamines by Unactivated Smiles Rearrangement of N-Aryl Anthranilamides
Romain Costil, Harvey J. A. Dale, Natalie Fey, George Whitcombe, Johnathan V. Matlock, Jonathan Clayden
Angew. Chemie Int. Ed. 2017, 56, 12533-12537 [doi 10.1002/anie.201706341]
263. Geometry-selective C-alkenylation of lithiated α-aminonitriles: quaternary α-alkenyl amino acids and hydantoins
Josep Mas-Roselló, Shuji Hachisu and Jonathan Clayden
Angew. Chemie Int. Ed. 2017, 56, 10750-10754 [doi 10.1002/anie.201704908]
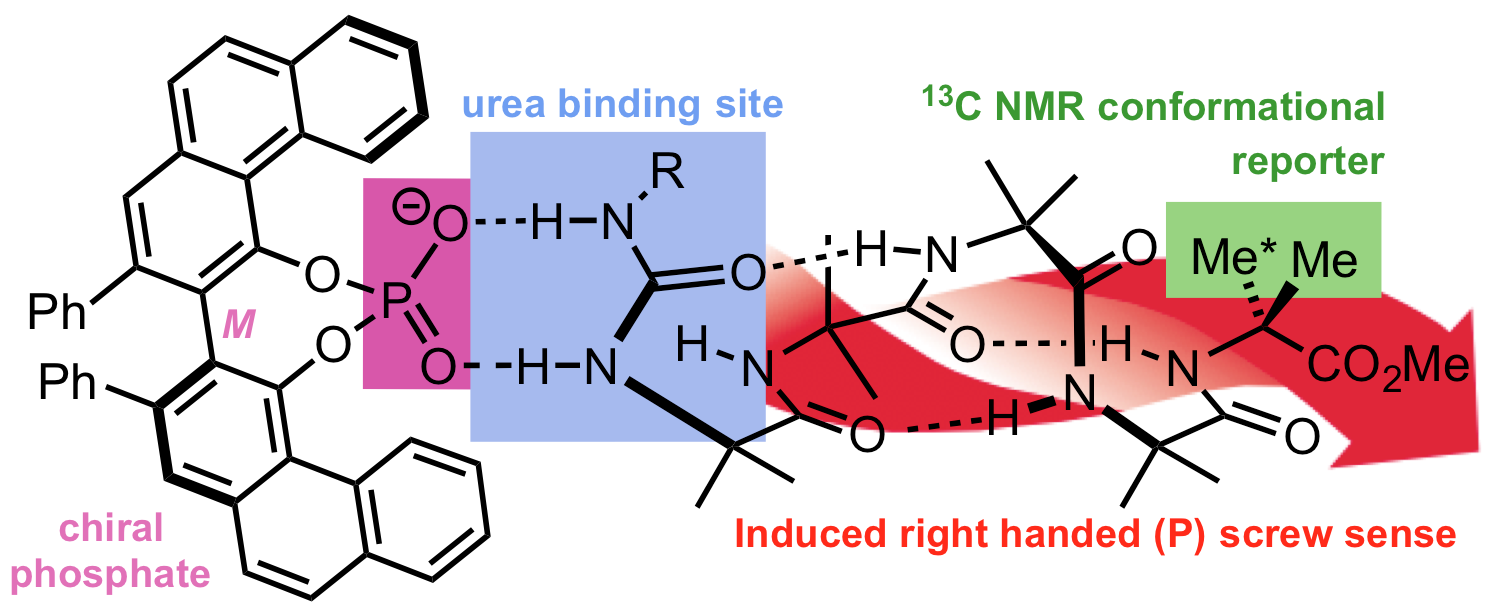
262. Signal transduction in oligoamide foldamers by selective non-covalent binding of chiral phosphates at a urea binding site
Katharina Gratzer, Vincent Diemer, Jonathan Clayden
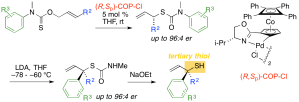
Org. Biomol. Chem. 2017, 15, 3585-3589 [doi 10.1039/C7OB00660H]
261. “Host in Translation”
Jonathan Clayden
Nature Nanotechnology, 2017, 12, 403 [News and views article] [doi 10.1038/nnano.2017.19]
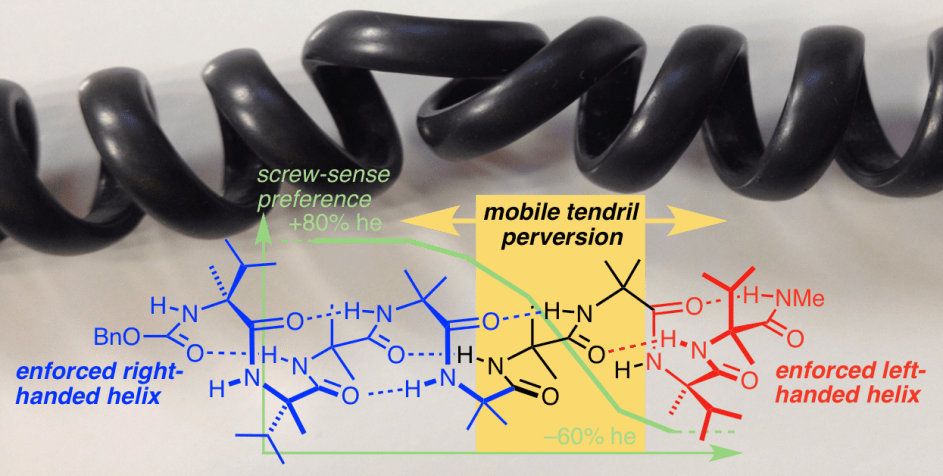
260. A tendril perversion in a helical oligomer: trapping and characterizing a mobile screw-sense reversal
Michael Tomsett, Irene Maffucci, Bryden A. F. Le Bailly, Liam Byrne, Stefan M. Bijvoets,
M. Giovanna Lizio, James Raftery, Craig P. Butts, Simon J. Webb, Alessandro Contini, Jonathan Clayden
Chem. Sci., 2017, 8, 3007-3018 [doi 10.1039/C6SC05474A]
259. Ligand-modulated conformational switching in a fully synthetic membrane-bound receptor
Francis G. A. Lister, Bryden A. F. Le Bailly, Simon J. Webb and Jonathan Clayden
Nature Chem., 2017, 9, 420-425 [doi 10.1038/nchem.2736].
See also the Nature Chemistry editorial ‘Sending a message to the other side’ (Nature Chem., 2017, 9, 403 [doi 10.1038/nchem.2776]) and Highlight article ‘Membrane messengers’ (Nature Chem., 2017, 9, 403 [doi 10.1038/nchem.2775]) covering this work.
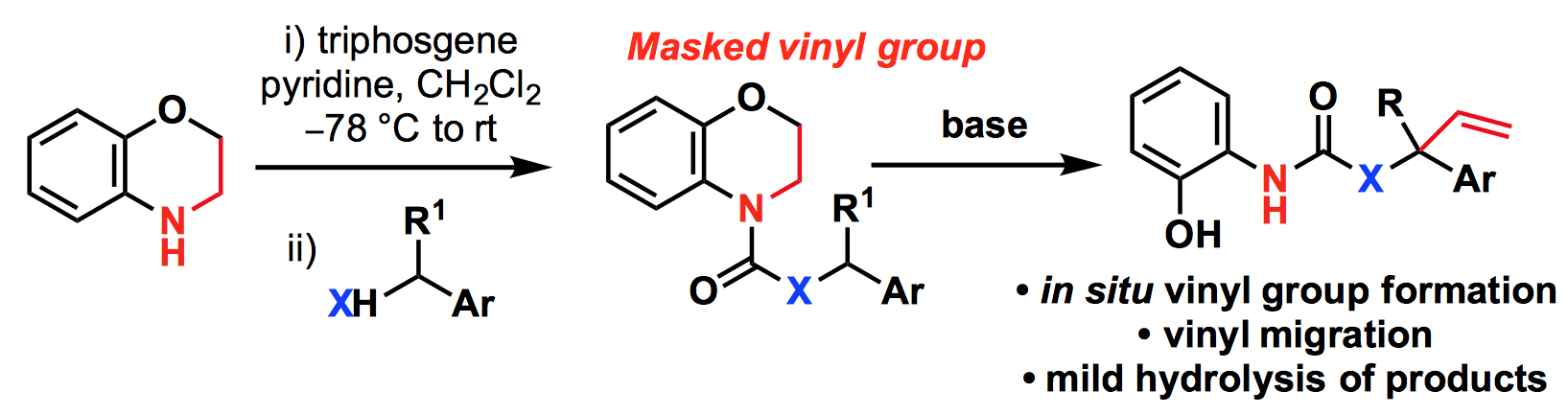
258. Intramolecular Vinylation of Carbanions Using N-Acyl Benzomorpholines as Masked Vinylureas and Vinylcarbamates / Vinylation intramoléculaire de carbanions nucléophiliques par les N-acylbenzomorpholines comme vinylurées et vinylcarbamates masqués
Brian P. Corbet, Johnathan V. Matlock, Josep Mas-Roselló, Jonathan Clayden
Comptes Rendus Chimie, 2017, 20, 634-642 [doi 10.1016/j.crci.2017.01.006] [Special issue on Carbanion Chemistry]
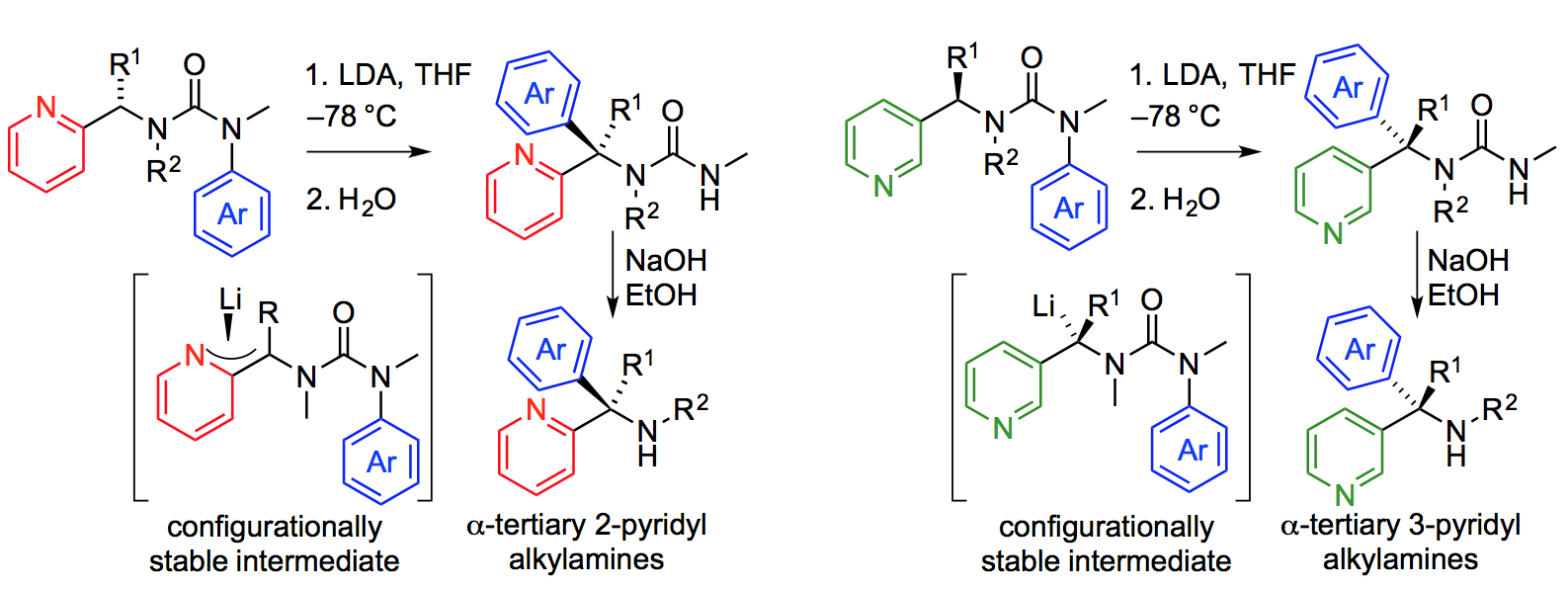
257. Stereospecific Intramolecular Arylation of 2- and 3-Pyridyl Substituted Alkylamines via Configurationally Stable alpha-Pyridyl Organolithiums
Julien Maury, Wojciech Zawodny, Jonathan Clayden
Org. Lett., 2017, 19, 472–475 [doi 10.1021/acs.orglett.6b03603]
2016
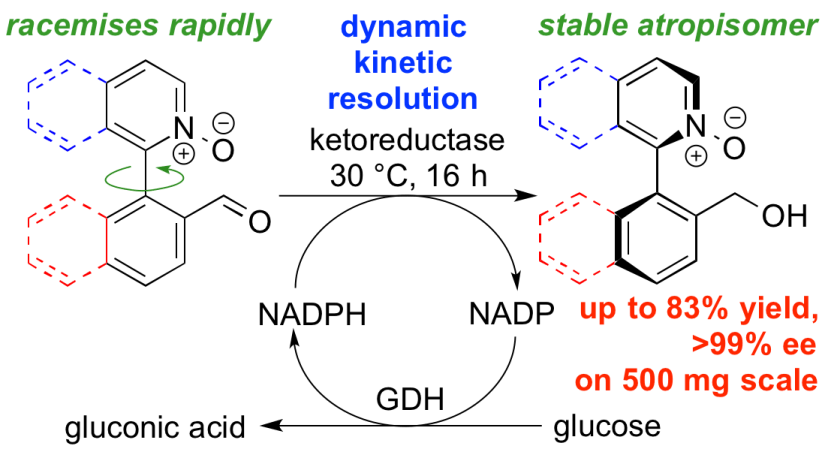
256. Biocatalytic Dynamic Kinetic Resolution for the Synthesis of Atropisomeric Biaryl N-Oxide Lewis Base Catalysts
Samantha Staniland, Ralph W. Adams, Joseph J. W. McDouall, Irene Maffucci, Alessandro Contini, Damian Grainger, Nicholas J. Turner, Jonathan Clayden
Angew. Chemie Int. Ed., 2016, 55, 10755-10759 [doi 10.1002/anie.201605486]
255. Medium Ring Nitrogen Heterocycles by Migratory Ring Expansion of Metallated Ureas
Jessica E. Hall, Johnathan V. Matlock, John W. Ward, Katharine V. Gray, Jonathan Clayden
Angew. Chemie Int. Ed., 2016, 55, 11153-11157 [doi 10.1002/anie.201605714] (‘VIP paper’ with ChemistryViews highlight feature and Synfacts report)

254. Helical foldamers incorporating photoswitchable residues for light-mediated modulation of conformational preference
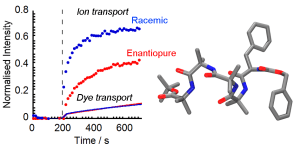
Daniela Mazzier, Marco Crisma, Matteo De Poli, Giulia Marafon, Cristina Peggion, Jonathan Clayden, Alessandro Moretto
J. Am. Chem. Soc., 2016, 138, 8007-8018 [doi 10.1021/jacs.6b04435]
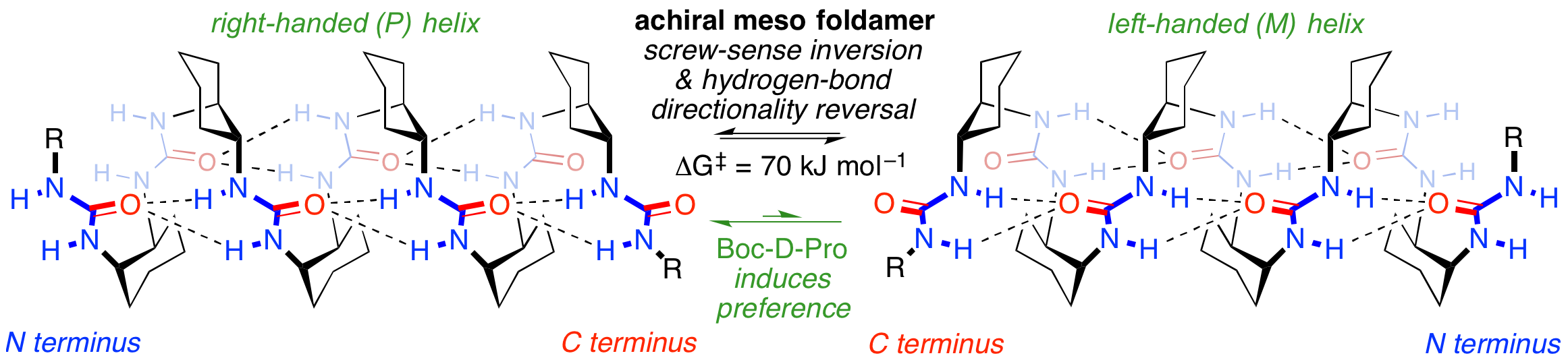
253. The meso helix: symmetry and symmetry-breaking in dynamic oligourea foldamers with reversible hydrogen-bond polarity
Romina Wechsel, James Raftery, Dominique Cavagnat, Gilles Guichard, Jonathan Clayden
Angew. Chemie Int. Ed., 2016, 55, 9657-9661 [doi 10.1002/anie.201604496] (‘Hot Paper’)
252. No turning back for motorized molecules
Jonathan Clayden
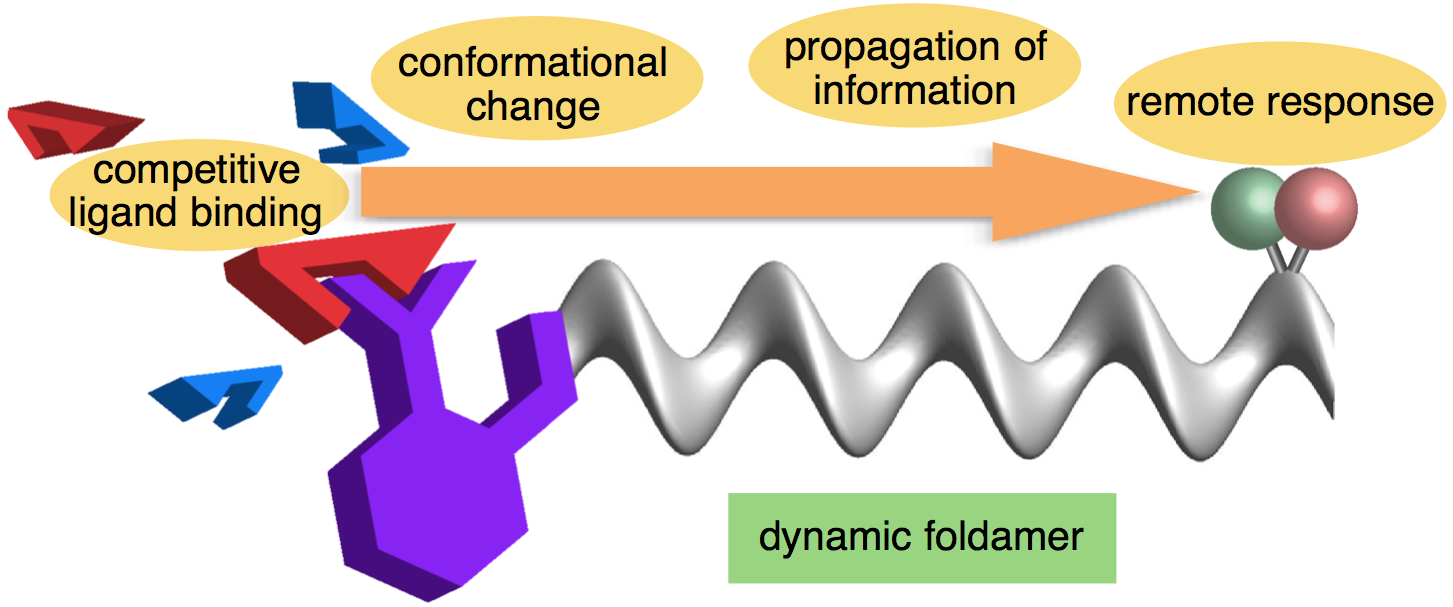
Nature, 2016, 534, 187-188 [News and views article] [doi:10.1038/534187a]

251. Dynamic foldamer chemistry
Bryden A. F. Le Bailly and Jonathan Clayden
Chem. Commun., 2016, 52, 4852-4863 [review] [doi 10.1039/C6CC00788K]
250. Conformational photoswitching of a synthetic peptide foldamer bound within a phospholipid bilayer
Matteo De Poli, Wojciech Zawodny, Ophélie Quinonero, Mark Lorch, Simon J. Webb and Jonathan Clayden
Science 2016, 352, 575-580 [doi 10.1126/science.aad8352]
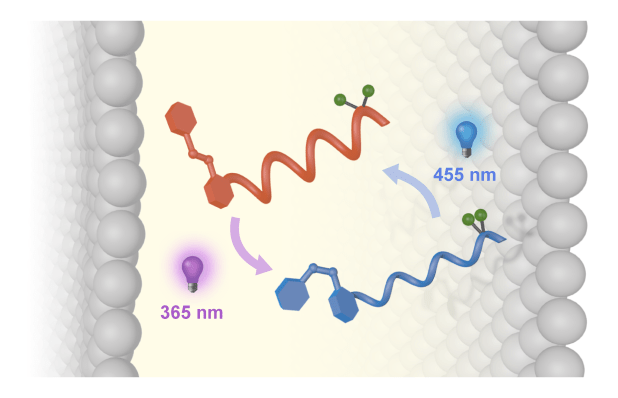
See also Insights summary article: C. M. Thiele, A. S. Ulrich, Science 2016, 352, 520 [doi 10.1126/science.aaf6191]
249. Substituent effects on axial chirality in 1-aryl-3,4-dihydroisoquinolines: controlling the rate of bond rotation
Josep Mas Roselló, Samantha Staniland, Nicholas J. Turner and Jonathan Clayden
Tetrahedron 2016, 72, 5172-5177 (Symposium in print ‘Control of Axial Chirality’) [doi 10.1016/j.tet.2016.01.037]
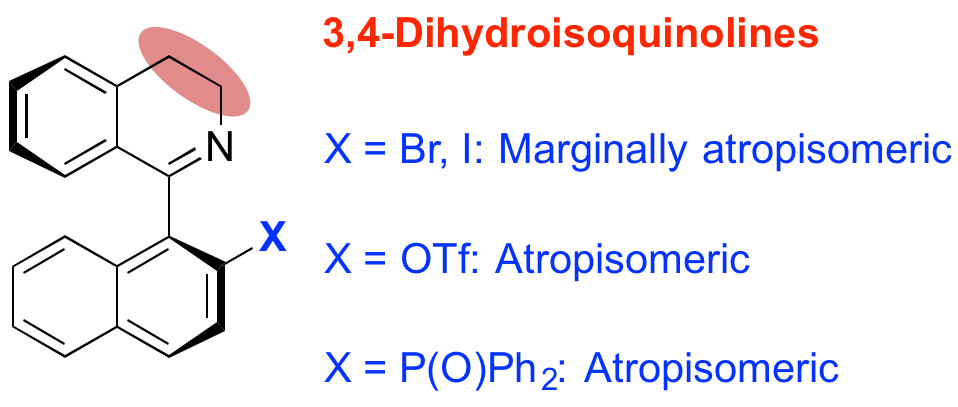
248. Length-dependent formation of transmembrane pores by 310 helical Aib foldamers
Jennifer E. Jones, Vincent Diemer, Catherine Adam, James Raftery, Rebecca E. Ruscoe, Jason Sengel, Mark I. Wallace, Antoine Bader, Scott L. Cockroft, Jonathan Clayden and Simon J. Webb
J. Am. Chem. Soc. 2016, 132, 688-695 [doi 10.1021/jacs.5b12057]
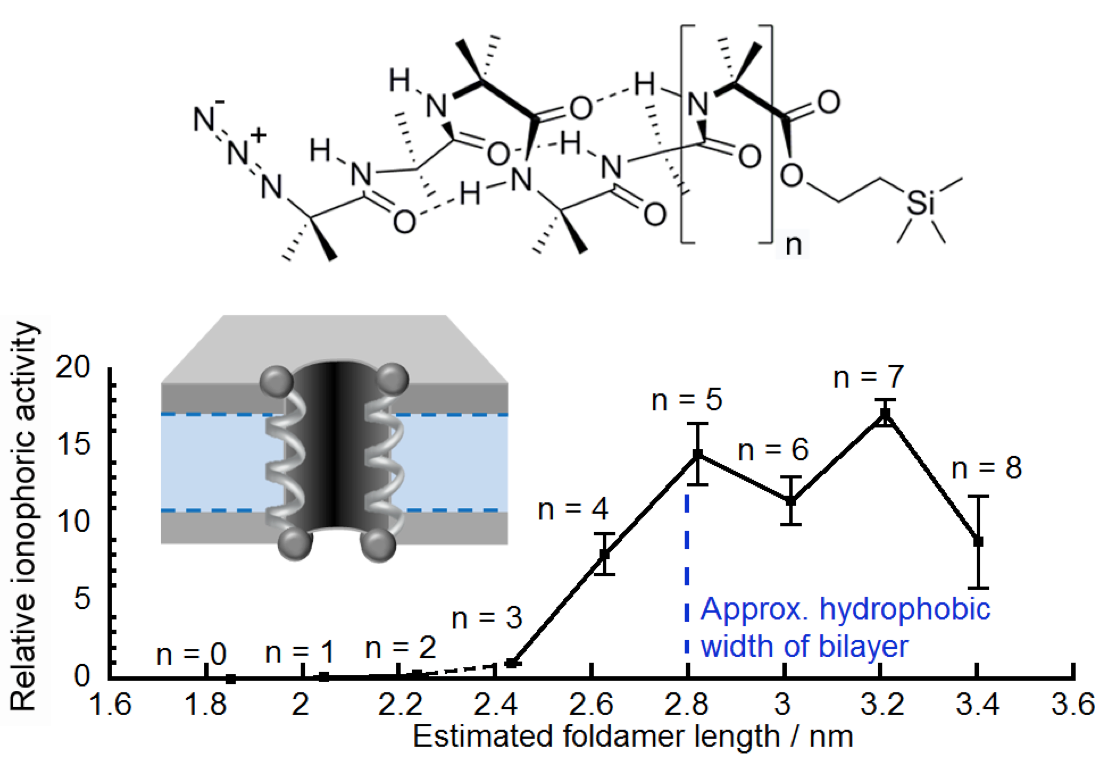
247. Refoldable foldamers: global conformational switching by deletion or insertion of a single hydrogen bond
Bryden A. F. Le Bailly, Liam Byrne and Jonathan Clayden
Angew. Chemie Int. Ed. 2016, 55, 2312-2316 [doi 10.1002/anie.201510605]

2015
246. Origin of Helical Screw Sense Selectivity Induced by Chiral Constrained Cα-Tetrasubstituted α-Amino Acids in Aib-based Peptides
Irene Maffucci, Jonathan Clayden and Alessandro Contini
J. Phys. Chem. B 2015, 119, 14003-14013 [doi 10.1021/acs.jpcb.5b07050]
245. α-Quaternary proline derivatives by intramolecular diastereoselective arylation of N-carboxamido proline ester enolates
Julien Maury and Jonathan Clayden
J. Org. Chem. 2015, 80, 10757-10768 [doi 10.1021/acs.joc.5b01912]
244. Helical peptaibol mimics are better ionophores when racemic than when enantiopure
Sarah J. Pike, Jennifer E. Jones, James Raftery, Jonathan Clayden, and Simon J. Webb
Org. Biomol. Chem. 2015, 13, 9580-9584 [doi 10.1039/C5OB01652E
243. Palladium-catalysed C-arylation of amino acid derived hydantoins
Fernando Fernández-Nieto, Josep Mas Roselló, Simone Lenoir, Simon Hardy and Jonathan Clayden
Org. Lett. 2015, 17, 3838-3841 [doi 10.1021/acs.orglett.5b01803]
242. Conformational cooperativity between helical domains of differing geometry in oligoamide-oligourea foldamer chimeras
Julien Maury, Bryden A. F. Le Bailly, James Raftery and Jonathan Clayden
Chem. Commun. 2015, 51, 11802-11805 [doi 10.1039/C5CC02995C]
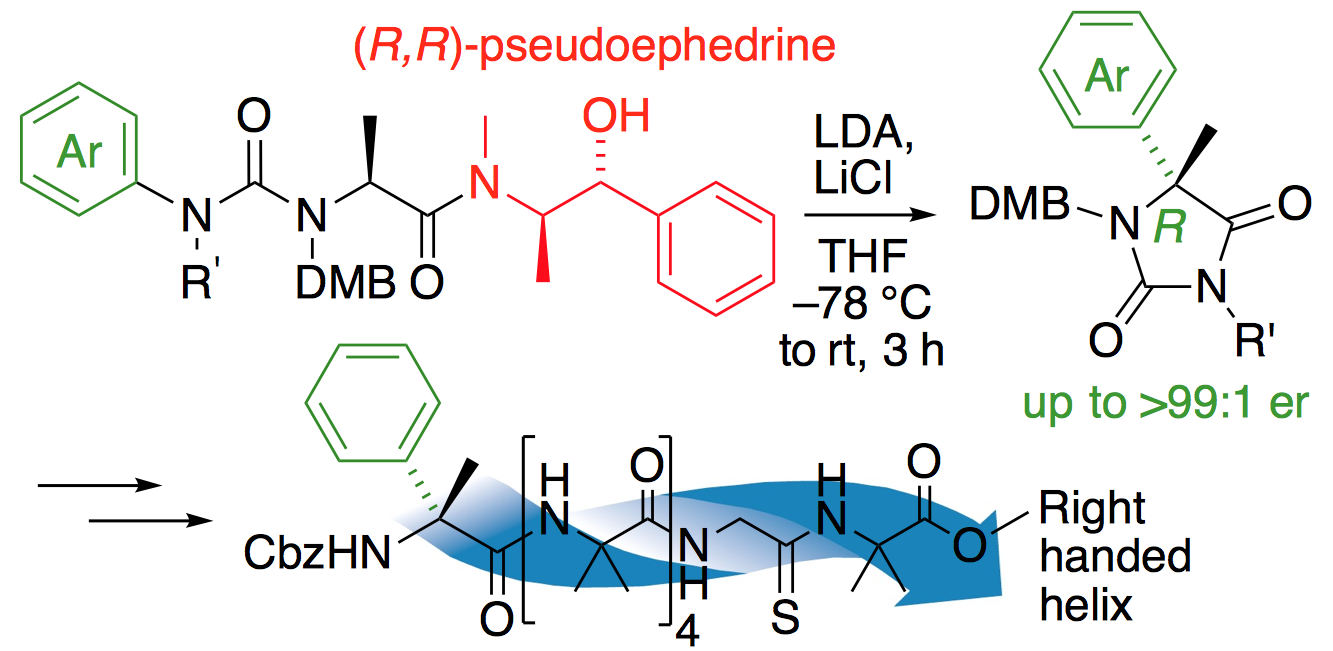
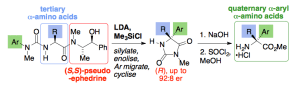
241. Pseudoephedrine-directed asymmetric alpha-arylation of alpha-amino acid derivatives
Rachel C. Atkinson, Fernando Fernández-Nieto, Josep Mas Roselló and Jonathan Clayden
Angew. Chemie Int. Ed. 2015, 54, 8961-8965 [doi 10.1002/anie.201502569]
240. Conformational switching of a foldamer in a multi-component system by pH-filtered selection between competing non-covalent interactions
Julien Brioche, Sarah J. Pike, Sofja Tshepelevitsh, Ivo Leito, Gareth A. Morris, Simon J. Webb, and Jonathan Clayden
J. Am. Chem. Soc. 2015, 137, 6680-6691 [doi 10.1021/jacs.5b03284]
239. Screw sense alone can govern enantioselective extension of a helical peptide by kinetic resolution of a racemic amino acid
Liam Byrne, Jordi Solà and Jonathan Clayden
Chem. Commun. 2015, 51, 10965-10968 [doi 10.1039/c5cc01790d]
238. Geometry-selective synthesis of the unsaturated side chains of the isodomoic acids
Nadia Fleary-Roberts, Gilles Lemière and Jonathan Clayden
Tetrahedron 2015, 71, 7204-7208 (Alan Katritzky memorial issue) [doi 10.1016/j.tet.2015.02.055]
237. 2,2- And 2,6-diarylpiperidines by aryl migration within lithiated urea derivatives of tetrahydropyridines
Michael B. Tait, Sam Butterworth, and Jonathan Clayden
Org. Lett. 2015, 17, 1236-1239 [doi 10.1021/acs.orglett.5b00199]
236. Flaws in foldamers: conformational uniformity and signal decay in achiral helical peptide oligomers
Bryden A. F. Le Bailly, Liam Byrne, Vincent Diemer, Mohammadali Foroozandeh, Gareth A. Morris, and Jonathan Clayden
Chem. Sci. 2015, 6, 2313-2322 [doi 10.1039/C4SC03944K]
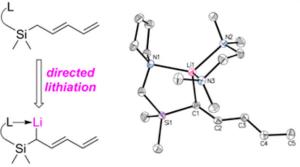
235. Directed lithiation of pentadienylsilanes
Benjamin M. Day, Joseph J. W. McDouall, Jonathan Clayden, and Richard A. Layfield
Organometallics 2015, 34, 2348-2355 [doi 10.1021/om501144f]
234. Recent developments in inter- and intra-molecular enolate arylation
Fernando Fernández-Nieto and Jonathan Clayden
Reference Module in Chemistry, Molecular Sciences and Chemical Engineering (ed. J. Reedijk) Elsevier, Oxford (ISBN 9780124095472) [doi 10.1016/B978-0-12-409547-2.11043-1]
233. Enantioselective carbolithiation of S-alkenyl-N-aryl thiocarbamates: kinetic and thermodynamic control
Daniele Castagnolo, Leonardo Degennaro, Renzo Luisi, and Jonathan Clayden
Org. Biomol. Chem. 2015, 13, 2330-2340 [doi 10.1039/C4OB02329C]
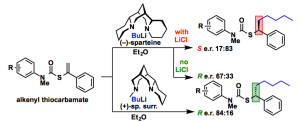
232. Lithium choreography determines contrasting stereochemical outcomes of aryl migrations in benzylic carbamates, ureas and thiocarbamates
Mark A. Vincent, Julien Maury, Ian H. Hillier and Jonathan Clayden
Eur. J. Org. Chem. 2015, 953-959 [doi 10.1002/ejoc.201403572]
231. Participation of non-aminoisobutyric acid (Aib) residues in the 310 helical conformation of Aib-rich foldamers: a solid state study
Sarah J. Pike, Thomas Boddaert, James Raftery, Simon J. Webb and Jonathan Clayden
New J. Chem. 2015, 39, 3288-3294 (Special issue on Foldamers) [doi 10.1039/C4NJ01547A]
230. Mechanism of stabilization of helix secondary structure by constrained Cα-tetrasubstituted amino acids
Irene Maffucci, Sara Pellegrino, Jonathan Clayden and Alessandro Contini
J. Phys. Chem. B 2015, 119, 1350-1361 [doi 10.1021/jp510775e]
2014
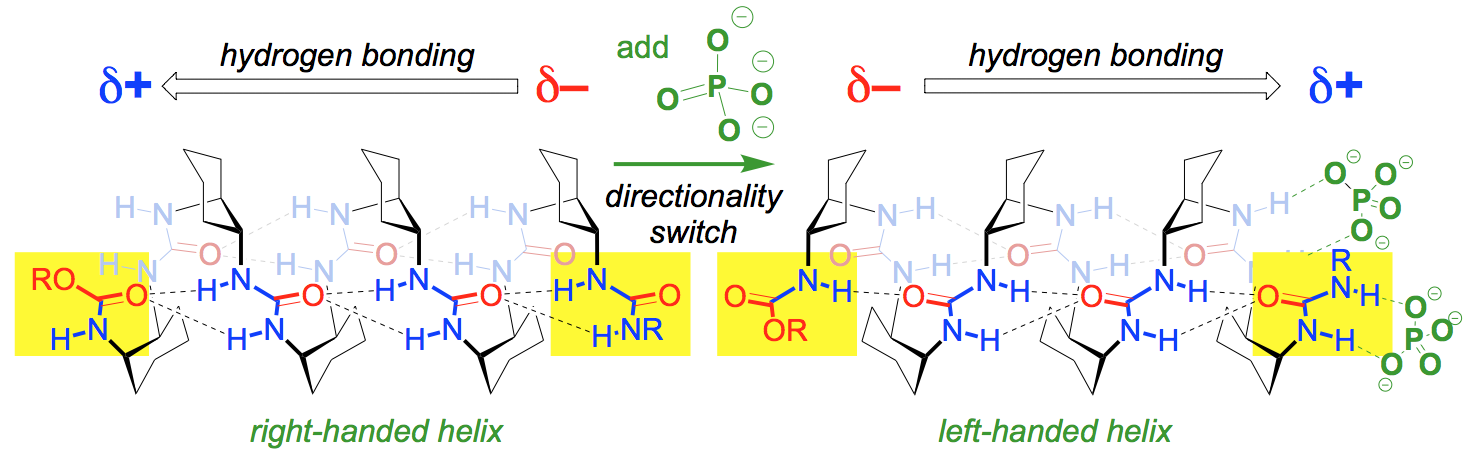
229. Inducing achiral aliphatic oligoureas to fold into helical conformations
Romina Wechsel, Julien Maury, Juliette Fremaux, Scott P. France, Gilles Guichard and Jonathan Clayden
Chem. Commun. 2014, 50, 15006-15009 [doi 10.1039/c4cc06754a]
228. Designing foldamer-foldamer interactions in solution: the roles of helix length and terminus functionality in promoting the self-association of aminoisobutyric acid oligomers
Sarah J. Pike, Vincent Diemer, James Raftery, Simon J. Webb, Jonathan Clayden
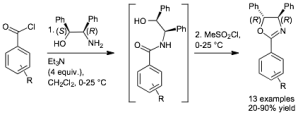
Chem. Eur. J. 2014, 20, 15981-15990 [doi 10.1002/chem.201403626]
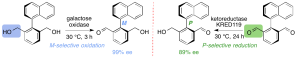
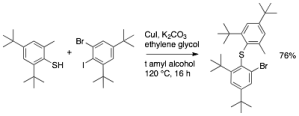
227. Enzymatic desymmetrising redox reactions for the asymmetric synthesis of biaryl atropisomers
Samantha Staniland, Bo Yuan, Nelson Giménez-Agulló, Tommaso Marcelli, Simon Willies, Damian Grainger, Nicholas J. Turner and Jonathan Clayden
Chem. Eur. J. 2014, 20, 13084-13088 [doi 10.1002/chem.201404509]
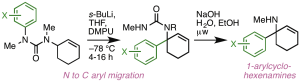
226. Synthesis of 1-arylcycloalkenamines by intramolecular arylation of lithiated ureas
Michael B. Tait, Philipp A. Ottersbach, Daniel J. Tetlow, and Jonathan Clayden
Org. Proc. Res. Dev. 2014, 18, 1245-1252 [doi 10.1021/op500173q]
225. Atropisomerism about Aryl−Csp3 Bonds: The Electronic and Steric Influence of ortho-Substituents on Conformational Exchange in Cannabidiol and Linderatin Derivatives
Hatice Berber, Pedro Lameiras, Clément Denhez, Cyril Antheaume and Jonathan Clayden
J. Org. Chem. 2014, 79, 6015-6027 [doi 10.1021/jo5006069]
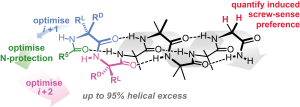
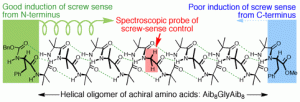
224. Controlling the sign and magnitude of screw-sense preference from the C-terminus of an achiral helical foldamer
Bryden A. F. Le Bailly and Jonathan Clayden
Chem. Commun. 2014, 50, 7949-7952 [doi 10.1039/c4cc03261f]
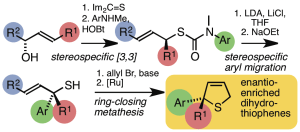
223. Dihydrothiophenes containing quaternary stereogenic centres by sequential stereospecific rearrangements and ring-closing metathesis
Gaëlle Mingat, Joseph J. W. McDouall and Jonathan Clayden
Chem. Commun. 2014, 50, 6754-6757 [doi 10.1039/c4cc02596b]
222. Engineering the structure of an N-terminal beta-turn to maximize screw-sense preference in achiral helical peptide chains
Matteo De Poli, Liam Byrne, Robert A. Brown, Jordi Solà, Alejandro Castellanos, Thomas Boddaert, Romina Wechsel, Jonathan D. Beadle, and Jonathan Clayden
J. Org. Chem. 2014, 79, 4659-4675 [doi 10.1021/jo500714b]
221. Conformational analysis of helical aminoisobutyric acid (Aib) oligomers bearing C-terminal schellman motifs
Sarah J. Pike, Simon Webb and Jonathan Clayden
Org. Biomol. Chem. 2014, 12, 4124-4131 [doi 10.1039/c4ob00268g]
220. Comprehensive Organic Synthesis II, vol. 8 (Reduction)
Jonathan Clayden (ed.)
in Comprehensive Organic Synthesis II, ed. P. Knochel and G. A. Molander, pub. Elsevier 2014, vol. 8.
219. Dearomatization and aryl migration in organolithium chemistry
Jonathan Clayden
in Lithium Compounds in Organic Synthesis, ed R. Luisi and V. Capriati, pub. Wiley-VCH 2014, chapter 13, pp. 375-394.
218. Tertiary thiols from allylic thiocarbamates by tandem enantioselective [3,3]-sigmatropic rearrangement and stereospecific arylation
Gaëlle Mingat, Paul MacLellan, Marju Laars and Jonathan Clayden
Org. Lett. 2014, 16, 1252-1255 [doi 10.1021/ol5002522]
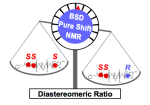
217. Diastereomeric Ratio Determination by High Sensitivity Band-Selective Pure Shift NMR Spectroscopy
Ralph W. Adams, Liam Byrne, Péter Király, Mohammadali Foroozandeh, Liladhar Paudel, Mathias Nilsson, Jonathan Clayden and Gareth A. Morris
Chem Commun. 2014, 50, 2512-2514 [doi 10.1039/c3cc49659g]
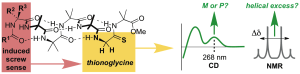
216. Thionoglycine as a multifunctional spectroscopic reporter of screw-sense preference in helical foldamers
Matteo De Poli and Jonathan Clayden
Org. Biomol. Chem. 2014, 12, 836-843 [doi 10.1039/c3ob42167h]
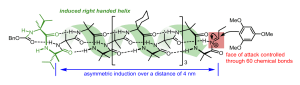
215. Foldamer-mediated remote stereocontrol: >1,60 asymmetric induction
Liam Byrne, Jordi Solà, Thomas Boddaert, Tommaso Marcelli, Ralph W. Adams, Gareth A. Morris and Jonathan Clayden
Angew. Chemie. Int. Ed. 2014, 53, 151-155 [doi 10.1002/anie.201308264] (‘VIP paper’)
See also Highlights summary article: C. P. Johnston and M. D. Smith Angew. Chem. Int. Ed. 2014, 53, 3315-3317 [doi 10.1002/anie.201400154]
2013
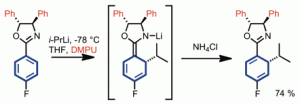
214. Intramolecular arylation of amino acid enolates
Rachel C. Atkinson, Daniel J. Leonard, Julien Maury, Daniele Castagnolo, Nicole Volz, and Jonathan Clayden
Chem. Commun. 2013, 49, 9734-9736 [doi 10.1039/c3cc46193a]
213. End-to-end conformational communication through a synthetic purinergic receptor by ligand-induced helicity switching
Robert A. Brown, Vincent Diemer, Simon J. Webb and Jonathan Clayden
Nature Chem. 2013, 5, 853-860 [doi 10.1038/nchem.1747]
212. The N-terminal nonapeptide of cephaibols A and C: a naturally occurring example of mismatched helical screw-sense control
Ugo Orcel, Matteo De Poli, Marta De Zotti and Jonathan Clayden
Chem. Eur. J. 2013, 19, 16357-16365 [doi 10.1002/chem.201302648]
211. Structural influences in lithium pentadienylsilane complexes
Benjamin M. Day, Jonathan Clayden and Richard A. Layfield
Organometallics 2013, 32, 4448-4451 [doi 10.1021/om400596y]
210. Solutions manual for Organic Chemistry, 2nd edition
Jonathan Clayden and Stuart Warren
ISBN 978-0-19-966334-7, pub. OUP , Oxford (2013)
209. Manipulating the diastereoselectivity of ortholithiation in planar chiral ferrocenes
Simon A. Herbert, Dominic C. Castell, Jonathan Clayden and Gareth E. Arnott
Org. Lett. 2013, 15, 3334-3337 [doi 10.1021/ol4013734]
208. Influence of achiral units with gem-dimethyl substituents on the helical character of aliphatic oligoureas
Juliette Fremaux, Christel Dolain, Brice Kauffmann, Jonathan Clayden and Gilles Guichard
Chem. Commun. 2013, 49, 7415-7417 [doi 10.1039/c3cc40961a]
207. Dearomatising cyclisation of lithiated allyl phenyl ethers: the role of an oxazoline substituent
Rebecca A. Harvey, Ol’ga Karlubíková, Sean Parris and Jonathan Clayden
Tetrahedron Lett. 2013, 54, 4064-4066 [10.1016/j.tetlet.2013.05.098]
206. Carbolithiation of S-alkenyl-N-aryl thiocarbamates: carbanion arylation in a connective route to tertiary thiols
Daniele Castagnolo, Daniel J. Foley, Hatice Berber, Renzo Luisi, and Jonathan Clayden
Org. Lett. 2013, 15, 2116-2119 [doi 10.1021/ol400570r]
205. Diastereotopic fluorine substituents as 19F NMR probes of screw-sense preference in helical foldamers
Sarah J. Pike, Matteo De Poli, Wojciech Zawodny, James Raftery, Simon J. Webb and Jonathan Clayden
Org. Biomol. Chem. 2013, 11, 3168-3176 [doi 10.1039/c3ob40463c]
204. Spirocyclic dihydropyridines by electrophile-induced dearomatizing cyclization of N-alkenyl pyridinecarboxamides
Jemma Senczyszyn, Heloise Brice and Jonathan Clayden
Org. Lett. 2013, 15, 1922-1925 [doi 10.1021/ol400571j]
203. Carbolithiation of N-alkenyl ureas and N-alkenyl carbamates
Julien Lefranc, Alberto Minassi and Jonathan Clayden
Beilstein J. Org. Chem. 2013, 6, 628-632 [doi 10.3762/bjoc.9.70]
202. Left-handed helical preference in an achiral peptide chain is induced by an L-amino acid in an N-terminal Type II beta-turn
Matteo De Poli, Marta De Zotti, James Raftery, Juan A. Aguilar, Gareth A. Morris and Jonathan Clayden
J. Org. Chem. 2013, 78, 2248-2255 [doi 10.1021/jo302705k]
201. Reversible aryl migrations in metallated ureas: controlled inversion of configuration at a quaternary carbon atom
Daniel J. Tetlow, Mark A. Vincent, Ian H. Hillier and Jonathan Clayden
Chem. Comm. 2013, 49, 1548-1550 [doi 10.1039/c2cc38704b]
200. Amines bearing tertiary substituents by tandem enantioselective carbolithiation–rearrangement of vinyl ureas
Michael Tait, Morgan Donnard, Alberto Minassi, Julien Lefranc, Beatrice Bechi, Giorgio Carbone, Peter O’Brien, and Jonathan Clayden
Org. Lett. 2013, 15, 34-37 [doi 10.1021/ol3029324] [corrigendum doi 10.1021/ol4002009]
2012
199. Lithium choreography: intramolecular arylations of carbamate-stabilised carbanions and their mechanisms probed by in-situ IR and DFT
Anne M. Fournier, Christopher J. Nichols, Mark A. Vincent, Ian H. Hillier and Jonathan Clayden
Chem. Eur. J. 2012, 18, 16478-16490 [doi 10.1002/chem.201201761]
198. S-Allyl thiocarbamates from allylic alcohols by in situ [3,3]-sigmatropic rearrangement of a thiocarbonyldiimidazole adduct
Gaëlle Mingat and Jonathan Clayden
Synthesis, 2012, 2723 [doi 10.1055/s-0032-1316746]
197. Lithiated tertiary carbanions display variable coordination modes: the evidence from DFT and NMR studies
Mark A. Vincent, Alison Campbell Smith, Morgan Donnard, Philip J. Harford, Joanna Haywood, Ian H. Hillier, Jonathan Clayden, and Andrew E. H. Wheatley
Chem. Eur. J. 2012, 18, 11036-11045 [doi 10.1002/chem.201200734]
196. Organic Chemistry (Second Edition)
Jonathan Clayden, Nick Greeves, and Stuart Warren
ISBN 0-19-927029-3, pub. OUP, Oxford (2012)
195. Intramolecular vinylation of secondary and tertiary organolithiums
Julien Lefranc, Anne M. Fournier, Gaëlle Mingat, Simon Herbert, Tommaso Marcelli, and Jonathan Clayden
J. Am. Chem. Soc. 2012, 134, 7286-7289 [doi 10.1021/ja301591m]
194. Chemical communication: conductors and insulators of screw-sense preference between helical oligo(aminoisobutyric acid) domains
Thomas Boddaert, Jordi Solà, Madeleine Helliwell and Jonathan Clayden
Chem. Commun. 2012, 48, 3397-3399 [doi 10.1039/c2cc00060a]
193. Stabilizers cause instability (News and Views article)
Jonathan Clayden
Nature (London) 2012, 481, 274-275 [doi 10.1038/481274a]
192. Induction of unexpected left-handed helicity by an N-terminal L-amino acid in an otherwise achiral peptide chain
Robert A. Brown, Tommaso Marcelli, Matteo De Poli, Jordi Solà, and Jonathan Clayden
Angew. Chemie Int. Ed. 2012, 51, 1395-1399 [doi 10.1002/anie.201107583]
191. Tertiary alcohols by tandem beta-carbolithiation and N to C aryl migration in enol carbamates
Anne M. Fournier and Jonathan Clayden
Org. Lett. 2012, 14, 142-145 [doi 10.1021/ol2029355]
190. On the control of secondary carbanion structure utilizing ligand effects during directed metalation
Andrew E. H. Wheatley, Jonathan Clayden, Ian H. Hillier, Alison Campbell Smith, Mark A. Vincent, Laurence J. Taylor and Joanna Haywood
Beilstein J. Org. Chem. 2012, 8, 50-60 [doi 10.3762/bjoc.8.5]
189. The Mechanism of the Stereospecific Intramolecular Arylation of Lithiated Ureas: The Role of Li+ probed by electronic structure calculations, and by NMR and IR spectroscopy
Damian M. Grainger, Alison Campbell Smith, Mark A. Vincent, Ian H Hillier, Andrew E. H. Wheatley and Jonathan Clayden
Eur. J. Org. Chem. 2012, 731-743 [doi 10.1002/ejoc.201101475]
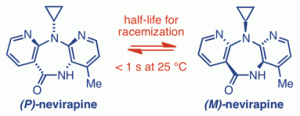
188. Is nevirapine atropisomeric? Experimental and computational evidence for rapid conformational inversion
Edmund W. D. Burke, Gareth A. Morris, Mark A. Vincent, Ian H. Hillier and Jonathan Clayden
Org. Biomol. Chem. 2012, 10, 716-719 [doi 10.1039/c1ob06490h]
2011
187. The Urea Renaissance
Nicole Volz and Jonathan Clayden
Angew. Chemie Int. Ed. 2011, 50, 12148-12155 [doi 10.1002/anie.201104037]
186. Dearomatizing reactions using organolithiums
Gilles Lemière and Jonathan Clayden
Science of Synthesis, Knowledge Updates 2011, 4, 139-190.
185. Communicating chirality (News and Views article)
Jonathan Clayden
Nature Chemistry 2011, 3, 842 [doi 10.1038/nchem.1181]
184. Carbamate-directed benzylic lithiation for the diastereo- and enantioselective synthesis of diaryl ether atropisomers
Abigail Page and Jonathan Clayden
Beilstein J. Org. Chem. 2011, 7, 1327-1333 [doi 10.3762/bjoc.7.156]
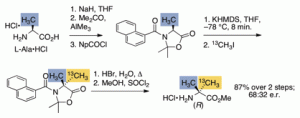
183. Synthesis of enantiomerically enriched (R)-13C-labelled 2-aminoisobutyric acid (Aib) by conformational memory in the alkylation of a derivative of L-alanine
Stephen P. Fletcher, Jordi Solà, Dean Holt, Robert A. Brown and Jonathan Clayden
Beilstein J. Org. Chem. 2011, 7, 1304-1309 [doi 10.3762/bjoc.7.152]
182. Ligand effects in the formation of tertiary carbanions from substituted tertiary aromatic amides
Alison Campbell Smith, Morgan Donnard, Joanna Haywood, Mary McPartlin, Mark A. Vincent, Ian H. Hillier, Jonathan Clayden, and Andrew E. H. Wheatley
Chem. Eur. J. 2011, 17, 8078-8084 [doi 10.1002/chem.201100240]
181. Attack on fluorinated 2-aryloxazolines by organolithiums: dearomatisation, lithiation or substitution
James Clayton and Jonathan Clayden
Tetrahedron Lett. 2011, 52, 2436-2439 [doi 10.1016/j.tetlet.2011.02.091]
180. Asymmetric synthesis of tertiary thiols and thioethers
Jonathan Clayden and Paul MacLellan
Beilstein J. Org. Chem. 2011, 7, 582-595 [doi 10.3762/bjoc.7.68]
179. Measuring screw-sense preference in a helical oligomer by comparison of 13C NMR signal separation at slow and fast exchange
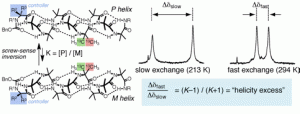
Jordi Solà, Gareth A. Morris and Jonathan Clayden
J. Am. Chem. Soc. 2011, 133, 3712-3715 [doi 10.1021/ja1097034]
178. Quaternary centres bearing nitrogen (alpha-tertiary amines) as products of molecular rearrangements
Jonathan Clayden, Morgan Donnard, Julien Lefranc and Daniel J. Tetlow
Chem. Commun. 2011, 4624-4639 [doi 10.1039/c1cc00049g]
177. A general synthetic approach to the amnesic shellfish toxins: total synthesis of (–)-isodomoic acid B, (–)-isodomoic acid E and (–)-isodomoic acid F
Gilles Lemière, Simon Sedehizadeh, Julie Toueg, Nadia Fleary-Roberts and Jonathan Clayden
Chem. Commun. 2011, 3745-3747 [doi 10.1039/C1CC00048A]
176. Enantioselective synthesis of tertiary thiols by intramolecular arylation of lithiated thiocarbamates
Paul MacLellan and Jonathan Clayden
Chem Commun. 2011, 3395-3397 [doi 10.1039/c0cc04912c]
175. Geometry-selective synthesis of E or Z N-vinyl ureas (N-carbamoyl enamines)
Julien Lefranc, Daniel J. Tetlow, Morgan Donnard, Alberto Minassi, Erik Gálvez and Jonathan Clayden
Org. Lett. 2011,13, 296-299 [doi 10.1021/ol1027442]
174. Interruption of a 310-helix by single Gly residue in a poly-Aib motif: a crystallographic study
Jordi Solà, Madeleine Helliwell and Jonathan Clayden
Biopolymers 2011, 95, 62-69 [doi 10.1002/bip.21535]
2010
173. Sequential double alpha-arylation of N-allylureas by asymmetric deprotonation and N to C aryl migration
Daniel J. Tetlow, Ulrich Hennecke, James Raftery, Michael J. Waring, David S. Clarke and Jonathan Clayden
Org. Lett. 2010, 12, 5442-5445 [doi 10.1021/ol102155h]
172. The origin of the conformational preference of N,N’-diaryl-N,N’-dimethyl ureas
Jonathan Clayden, Ulrich Hennecke, Mark A. Vincent, Ian H. Hillier and Madeleine Helliwell
Phys. Chem. Chem. Phys. 2010, 12, 15056-15064 [doi 10.1039/c0cp00571a]
171. Biocatalytic desymmetrisation of an atropisomer mediated by both an enantioselective oxidase and ketoreductases
Bo Yuan, Abigail Page, Christopher P. Worrall, Franck Escalettes, Simon C. Willies, Joseph J. W. McDouall, Nicholas J. Turner and Jonathan Clayden
Angew. Chemie Int. Ed. 2010, 49, 7010-7013 [doi 10.1002/anie.201002580]
170. Nanometre-range communication of stereochemical information by reversible switching of molecular helicity
Jordi Solà, Stephen P. Fletcher, Alejandro Castellanos and Jonathan Clayden
Angew. Chemie Int. Ed. 2010, 49, 6836-6839 [doi 10.1002/anie.201001130]
169. Deconstructing THF [news and views article]
Jonathan Clayden,
Nature Chemistry, 2010, 2, 523-524
168. Hindered diarylether and diarylsulfone bisphosphine ligands: atropisomerism and palladium complexes
Jonathan Clayden, Stephen P. Fletcher, James Senior and Christopher P. Worrall
Tetrahedron: Asymm. 2010, 21, 1355-1360 [doi 10.1016/j.tetasy.2010.06.017}
167. Conformational studies of tertiary oligo-m-benzanilides and oligo-p-benzanilides in solution
Laurent Chabaud, Jonathan Clayden, Madeleine Helliwell, Abigail Page, James Raftery and Lluís Vallverdú
Tetrahedron 2010, 66, 6936-6957 [doi 10.1016/j.tet.2010.06.037]
166. Tandem beta-alkylation–alpha-arylation of amines by carbolithiation and rearrangement of N-carbamoyl enamines (vinyl ureas)
Jonathan Clayden, Morgan Donnard, Julien Lefranc, Alberto Minassi and Daniel J. Tetlow
J. Am Chem. Soc. 2010, 132, 6624-6625 [doi 10.1021/ja1007992]
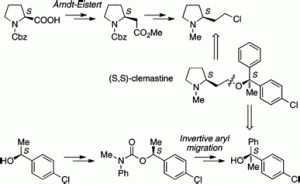
165. Synthesis of (–)-(S,S)-clemastine by invertive N to C aryl migration in a lithiated carbamate
Anne M. Fournier, Robert A. Brown, William Farnaby, Hideki Miyatake-Ondozabal, and Jonathan Clayden
Org. Lett. 2010, 12, 2222-2225 [doi 10.1021/ol100627c]
164. N- vs. C-terminal control over the screw-sense preference of the configurationally achiral, conformationally helical peptide motif Aib8GlyAib8
Jordi Solà, Madeleine Helliwell and Jonathan Clayden
J. Am. Chem. Soc. 2010, 132, 4548-4549 [doi 10.1021/ja100662d]
163. Fused bicyclic piperidines and dihydropyridines by dearomatising cyclisation of the enolates of nicotinyl-substituted esters and ketones
Heloise Brice, Jonathan Clayden and Stuart D. Hamilton
Beilstein J. Org. Chem. 2010, 6, No 22 [doi 10.3762/bjoc.6.22]
2009
162. A one-pot synthesis of 2-aryl-4,5-anti-diphenyl oxazolines
Jonathan Clayden, James Clayton, Rebecca A. Harvey, and Ol’ga Karlubíková
Synlett 2009, 2836-2838 [doi 10.1055/s-0029-1217985]
161. Direct synthesis of sulfonated azacalixarenes in water
Jonathan Clayden, Stephen J. M. Rowbottom, Warren J. Ebenezer and Michael G. Hutchings,
Org. Biomol. Chem. 2009, 4871-4880 [doi 10.1039/b911631a]
160. Synthesis of 2,2’,6-trisubstituted and 2,2’,6,6’-tetrasubstituted diaryl sulfides and diaryl sulfones by copper-promoted coupling and / or ortholithiation
Jonathan Clayden and James Senior
Synlett 2009, 2769-2772 [doi 10.1055/s-0029-1217986]
159. The challenge of atropisomerism in drug discovery
Jonathan Clayden, Wesley J. Moran, Paul J. Edwards and Steven R. LaPlante
Angew. Chemie Int. Ed. 2009, 48, 6398-6401 [doi 10.1002/anie.200901719]
158. Atropisomerism at C–S bonds: asymmetric synthesis of diaryl sulfones by dynamic resolution under thermodynamic control
Jonathan Clayden, James Senior and Madeleine Helliwell
Angew. Chemie Int. Ed. 2009, 48, 6270-6273 [doi 10.1002/anie.200901718]
157. Quantifying end-to-end conformational communication of chirality through an achiral peptide chain
Jonathan Clayden, Alejandro Castellanos, Jordi Solà and Gareth A. Morris
Angew. Chemie Int. Ed. 2009, 48, 5962-5965 [doi 10.1002/anie.200901892]
156. Conformational Preferences of a Polar Biaryl: a Phase- and Enantiomeric Purity-Dependent Molecular Hinge
Jonathan Clayden, Stephen P. Fletcher, S. J. M. Rowbottom and Madeleine Helliwell
Org. Lett. 2009, 11, 2313-2316 [doi 10.1021/ol9006635]
155. Formation of water-soluble sulfonated azacalix[4]arenes from cyanuric chloride
Jonathan Clayden, Stephen J. M. Rowbottom, Michael G. Hutchings and Warren J. Ebenezer
Tetrahedron Lett. 2009, 50, 3923-3925 [doi 10.1016/j.tetlet.2009.04.068]
154. Controlling axial conformation in 2-arylpyridines and 1-arylisoquinolines: application to the asymmetric synthesis of QUINAP by dynamic thermodynamic resolution
Jonathan Clayden, Stephen P. Fletcher, Joseph J. W. McDouall and Stephen J. M. Rowbottom
J. Am. Chem. Soc. 2009, 131, 5331-5434 [doi 10.1021/ja900722q]
153. N to C aryl migration in lithiated carbamates: alpha-arylation of benzylic alcohols
Jonathan Clayden, William Farnaby, Damian M. Grainger, Ulrich Hennecke, Michele Mancinelli, Daniel J. Tetlow, Ian H. Hillier and Mark A. Vincent
J. Am. Chem. Soc. 2009, 131, 3410-3411 [doi 10.1021/ja808959e]
152. Doubly dearomatising intramolecular coupling of a nucleophilic and an electrophilic heterocycle
Heloise Brice and Jonathan Clayden
Chem. Commun. 2009, 1964-1966 [doi 10.1039/b901558b]
151. Enantiomerically enriched atropisomeric N,N’-diaryl ureas by oxidative kinetic resolution of their 2-sulfanyl derivatives
Jonathan Clayden and Hazel Turner
Tetrahedron Lett. 2009, 50, 3216-3219 [doi 10.1016/j.tetlet.2009.02.021]
150. Transmission of stereochemical information over nanometre distances in chemical reactions
Jonathan Clayden
Chem. Soc. Rev. 2009, 38, 817-829 [doi 10.1039/b801639a]
149. alpha-Arylation of cyclic amines by aryl transfer in lithiated ureas
Renaud Bach, Jonathan Clayden and Ulrich Hennecke
Synlett, 2009, 421-424 [doi 10.1055/s-0028-1087543]
148. Relaying stereochemistry through aromatic ureas: 1,9 and 1,15 remote stereocontrol
Jonathan Clayden, Mark Pickworth and Lyn. H. Jones
Chem. Commun. 2009, 547-549 [doi 10.1039/b817527f]
2008
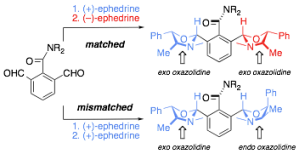
147. Asymmetric synthesis of biaryl atropisomers by dynamic resolution on condensation of biaryl aldehydes with (−)-ephedrine or a proline- derived diamine
Ann Bracegirdle, Jonathan Clayden and Lai Wah Lai
Beilstein J. Org. Chem. 2008, 4, 47 [doi 10.3762/bjoc.4.47]
146. Synthesis of enantiomerically enriched isotopically-labelled anilines by (–)-sparteine directed lithiation
Jonathan Clayden, Loïc Lemiègre and Mark Pickworth
Tetrahedron Asymmetry 2008, 19, 2218-2221 [doi 10.1016/j.tetasy.2008.09.014]
145. Helix persistence and breakdown in oligoureas of metaphenylenediamine: apparent diastereotopicity as a spectroscopic marker of helix length in solution
Jonathan Clayden, Loïc Lemiègre, Gareth A. Morris, Mark Pickworth, Timothy J. Snape and Lyn H. Jones
J. Am. Chem. Soc. 2008, 130, 15193-15202 [doi 10.1021/ja805758v]
144. alpha-Pyridylation of chiral amines via urea coupling, lithiation and rearrangement
Jonathan Clayden and Ulrich Hennecke [doi 10.1021/ol801332n]
Org. Lett. 2008, 10, 3567-3570
143. Conformation and Stereodynamics of 2,2’-Disubstituted N,N’-Diaryl Ureas
Jonathan Clayden, Loïc Lemiègre, Mark Pickworth and Lyn Jones
Org. Biomol. Chem. 2008, 6, 2908-2913 [doi 10.1039/b802673d]
142. Stereoselective Dearomatizing Addition of Nucleophiles to Uncomplexed Benzene Rings: A Route to Carbocyclic Sugar Analogues
Jonathan Clayden, Sean Parris, Nuria Cabedo and Andrew H. Payne
Angew. Chemie Int Ed. 2008, 47, 5060-5062 [doi 10.1002/anie.200801078]
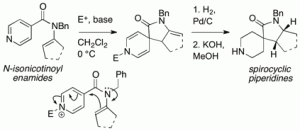
141. Electrophile-induced dearomatising spirocyclisation of N-aryl isonicotinamides: a route to spirocyclic piperidines
Gareth Arnott, Heloise Brice, Jonathan Clayden and Emma Blaney
Org. Lett. 2008, 10, 3089-3092 [doi 10.1021/ol801092s]
140. N,N’-Diarylureas: A New Family of Atropisomers Exhibiting Highly Diastereoselective Reactivity
Jonathan Clayden, Hazel Turner, Madeleine Helliwell and Elizabeth Moir
J. Org. Chem. 2008, 73, 4415-4423 [doi 10.1021/jo702706c]
139. Enantioselective synthesis of an atropisomeric diaryl ether
Jonathan Clayden, Christopher P. Worrall, Wesley J. Moran and Madeleine Helliwell
Angew. Chemie Int Ed. 2008, 47, 3234-3237 [doi 10.1002/anie.200705660]
138. Conformational switching between diastereoisomeric atropisomers of arenedicarboxamides induced by complexation with Lewis acids
Jonathan Clayden, Lluís Vallverdú, James Clayton and Madeleine Helliwell
Chem. Commun. 2008, 561-563 [doi 10.1039/b716105k]
2007
137. Remote Control of Stereochemistry: Communicating Information via Conformation
Jonathan Clayden, in Molecular Interactions – Bringing Chemistry to Life , ed. M. G. Hicks and C. Kettner, pub. Beilstein Institut 2007, ISBN 978-3-8325-1791-5.
136. Oxidative fragmentation of bicyclic hydroxy silanes and stannanes: a strategy for the stereoselective synthesis of kainoids
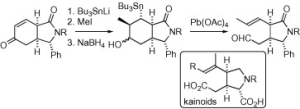
Jonathan Clayden, Katherine R. Hebditch, Benjamin Read and Madeleine Helliwell
Tetrahedron Lett. 2007, 48, 8550-8553 [doi 10.1016/j.tetlet.2007.09.150]
135. Substituted Diarylmethylamines by Stereospecific Intramolecular Electrophilic Arylation of Lithiated Ureas
Jonathan Clayden, Jérémy Dufour, Damian M. Grainger and Madeleine Helliwell
J. Am. Chem. Soc. 2007, 129, 7488-7489 [doi 10.1021/ja071523a]
134. Transmitting information along oligoparaphenylenes: 1,12-stereochemical control in a terphenyl tetracarboxamide
Jonathan Clayden, Lluís Vallverdú and Madeleine Helliwell
Chem. Commun. 2007, 2357-2359 [doi 10.1039/b701682d]
133. Synthesis of densely functionalised arenes using [2+2+2] cycloaddition reactions
Jonathan Clayden and Wesley J. Moran
Org. Biomol. Chem. 2007, 5, 1028-1030 [doi 10.1039/b700353f]
132. Synthesis and stacked conformations of symmetrical and unsymmetrical oligo-ureas of metaphenylenediamine
Jonathan Clayden, Loïc Lemiègre, and Madeleine Helliwell
J. Org. Chem. 2007, 72, 2302-2308 [doi 10.1021/jo061989w]
131. Achieving conformational control over C–C, C–N and C–O bonds in biaryls, N,N’-diarylureas and diaryl ethers: advantages of a relay axis
Mark S. Betson, Ann Bracegirdle, Jonathan Clayden, Madeleine Helliwell, Andrew Lund, Mark Pickworth, Timothy J. Snape and Christopher P. Worrall
Chem. Commun. 2007, 754-756 [doi 10.1039/b614618j]
2006
130. Azabicyclic amino acids by stereoselective dearomatizing cyclization of the enolates of N-nicotinoyl glycine derivatives
Gareth Arnott, Jonathan Clayden and Stuart D. Hamilton
Org. Lett. 2006, 8, 5325-5328 [doi 10.1021/ol062126s]
129. The twisted amide 2-quinuclidone: 60 years in the making
Jonathan Clayden and Wesley J. Moran
Angew. Chemie Int. Ed., 2006, 45, 7118-7120 [doi 10.1002/anie.200603016]
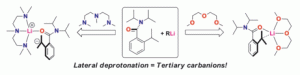
128. Lateral lithiation of N,N’-diaryl ureas
Jonathan Clayden and Jérémy Dufour
Tetrahedron Lett., 2006, 47, 6945-6946 [doi 10.1016/j.tetlet.2006.07.134]
127. BBC Radio 4: The Isomers Have It
Jonathan Clayden (presenter Sue Nelson; producer Helen Sharp), BBC Radio 4, 5th July 2006, 9.00-9.30 pm
126. Three groups good, four groups bad? Atropisomerism in ortho-substituted diaryl ethers
Mark S. Betson, Jonathan Clayden, Christopher P. Worrall and Simon Peace
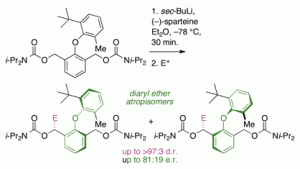
Angew. Chem. Int Ed., 2006, 45, 5803-5807 (Angew. Chemie 2006, 118, 5935) [doi 10.1002/anie.200601866]
125. Stereochemical relays: communication via conformation
Jonathan Clayden and Neoclis Vassiliou
Org. Biomol. Chem., 2006, 4, 2667-2678 [doi 10.1039/b604548k]
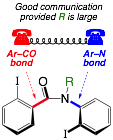
124. Conformational communication between the Ar–CO and Ar–N axes in 2,2’-disubstituted benzanilides and their derivatives
Jonathan Clayden, Lluís Vallverdú and Madeleine Helliwell
Org. Biomol. Chem. 2006, 4, 2106-2118 [doi 10.1039/b602912d]
123. Addition of lithiated tertiary aromatic amides to epoxides and aziridines: asymmetric synthesis of (S)-(+)-mellein
Jonathan Clayden, Christopher C. Stimson, Madeleine Helliwell and Martine Keenan
Synlett 2006, 873-876 [doi 10.1055/s-2006-939043]
122. Contra-Friedel-Crafts tert-butylation of substituted aromatic rings via directed metallation and sulfinylation
Jonathan Clayden, Christopher C. Stimson and Martine Keenan
Chem. Commun., 2006, 1393-1394 [doi 10.1039/b600181e]
121. Synthesis of multiply ortho-substituted diaryl ethers via lithiation and oxidation of a dibenzosiloxane (phenoxasilin)
Mark S. Betson and Jonathan Clayden
Synlett 2006, 745-746 [doi 10.1055/s-2006-933111]
120. Diastereoselective synthesis of atropisomers containing two non-biaryl stereogenic axes: stereochemical relay through stereogenic centres in dihydrostilbene-2,2’-dicarboxamides
Jonathan Clayden, Neil Westlund, Christopher S. Frampton and Madeleine Helliwell
Org. Biomol. Chem. 2006, 4, 455-461 [doi 10.1039/b514561a]
119. Conformational arm-wrestling: battles for stereochemical control in benzamides bearing matched and mismatched chiral 2- and 6- substituents
Jonathan Clayden, Yann J. Y. Foricher, Madeleine Helliwell, Paul Johnson, David Mitjans and Victoria Vinader
Org. Biomol. Chem. 2006, 4, 444-454 [doi 10.1039/b514558a]
118. Conformational preference in aromatic amides bearing chiral ortho substituents: its origin and application to relayed stereocontrol
Mark S. Betson, Jonathan Clayden, Madeleine Helliwell, Paul Johnson, Lai Wah Lai, Jennifer H. Pink, Christopher C. Stimson, Neoclis Vassiliou, Neil Westlund, Samreen A. Yasin and Latifa H. Youssef
Org. Biomol. Chem. 2006, 4, 424-443 [doi 10.1039/b514557k]
2005
117. Editorial – Inaugural issue of Beilstein Journal of Organic Chemistry
Jonathan Clayden
Beilstein. J. Org. Chem. 2005: 1.
116. Kinetic and thermodynamic stereocontrol in the atroposelective formation of sulfoxides by oxidation of 2-sulfanyl-1-naphthamides
Mark S. Betson, Jonathan Clayden, Madeleine Helliwell and David Mitjans
Org. Biomol. Chem. 2005, 3, 3898-3904 [doi 10.1039/b511452g]
115. Cyclization of lithiated pyridine- and quinolinecarboxamides: synthesis of partially saturated pyrrolopyridines and spirocyclic beta-lactams
Jonathan Clayden, Stuart D. Hamilton and Rukhsana T. Mohammed
Org. Lett. 2005, 7, 3673-3676 [doi 10.1021/ol051214u]
114. Slow interconversion of enantiomeric conformers or atropisomers of anilide and urea derivatives of 2-substituted anilines.
Thomas Adler, Josep Bonjoch, Jonathan Clayden, Mercè Font-Bardía, Mark Pickworth, Xavier Solans, Daniel Solé and Lluís Vallverdú
Org. Biomol. Chem. 2005, 3, 3173-3183 [doi 10.1039/b507202f]
113. Ring-selective functionalisation of N,N’-diarylureas by regioselective N-alkylation and directed ortho-metallation
Jonathan Clayden, Hazel Turner, Mark Pickworth and Thomas Adler
Org. Lett. 2005, 7, 3147-3151 [doi 10.1021/ol0508025]
112. Diastereoselective protonation of extended pyrrol-3-en-2-one enolates: an attempted “de-epimerisation”
Jonathan Clayden, Rachel Turnbull and Ivan Pinto
Tetrahedron: Asym. 2005, 16, 2235-2241 [doi 10.1016/j.tetasy.2005.05.042]
111. Asymmetric ortholithiation of amides by conformationally mediated chiral memory: an enantioselective route to naphtho- and benzofuranones
Jonathan Clayden, Christopher C. Stimson and Martine Keenan
Synlett 2005, 1716-1720 [doi 10.1055/s-2005-871554]
110. Focus Article: Ultra-remote stereocontrol by conformational communication of information along a carbon chain
Kenso Soai and Jonathan Clayden
Letters in Organic Chemistry, 2005, 2, 389-391.
109. Chemistry of domoic acid, isodomoic acids and their Analogues
Jonathan Clayden, Benjamin Read and Katherine R. Hebditch
Tetrahedron 2005, 61, 5713-5724 [doi 10.1016/j.tet.2005.04.003]
108. The synthesis of (–)-isodomoic acid C
Jonathan Clayden, Faye. E. Knowles and Ian R. Baldwin
J. Am. Chem. Soc. 2005, 127, 2412-2413 [doi 10.1021/ja042415g]
107. Using dipoles to control the directionality of functional groups: syn and anti oriented benzene-1,3-dicarboxamides
Mark S. Betson, Jonathan Clayden, Ho Kam Lam and Madeleine Helliwell
Angew. Chemie Int. Ed., 2005, 44, 1241-1244 (Angew. Chemie 2005, 117, 1267-1270) [doi 10.1002/anie.200461787]
106. Can relief of ring-strain in a cyclopropylmethyllithium drive the Brook rearrangement?
Jonathan Clayden, David W. Watson and Mark Chambers
Tetrahedron, 2005, 61, 3195 [doi 10.1016/j.tet.2004.10.099]
2004
105. ‡Book Review: “Stereochemistry at a glance”, J. Eames and J. Peach.
Jonathan Clayden
Chemistry World, Sept. 2004, 63
104. Dearomatising rearrangements of lithiated thiophenecarboxamides
Jonathan Clayden, Rachel Turnbull, Madeleine Helliwell and Ivan Pinto
Chem. Commun., 2004, 2430-2431 [doi 10.1039/b409150g]
103. Total synthesis of kainoids by dearomatizing anionic cyclisation
Jonathan Clayden
Strategies and Tactics in Organic Synthesis, vol. 4, p. 71. ed Michael Harmata, Academic Press, 2004
102. Cyclisations of organolithiums onto aromatic rings
Jonathan Clayden and Martin Kenworthy
Synthesis, 2004, 1721-1736 [doi 10.1055/s-2004-829138]
101. The directed metallation of aromatic compounds
Jonathan Clayden
in The Chemistry of Organolithium Compounds, pp 497-648, ed. Z. Rappoport and I. Marek, Wiley, Chichester, 2004
100. Ultra-remote stereocontrol by conformational communication of information along a carbon chain
Jonathan Clayden, Andrew Lund, Lluís Vallverdú and Madeleine Helliwell
Nature, 2004, 431, 966-971 [doi 10.1038/nature02933]
99. Dynamic resolution of atropisomeric amides using proline-derived imidazolidines and ephedrine-derived oxazolidines
Jonathan Clayden, Lai Wah Lai and Madeleine Helliwell
Tetrahedron, 2004, 60, 4399-4412 [doi 10.1016/j.tet.2004.01.101]
98. Sulfoxides as “traceless” resolving agents for the synthesis of atropisomers by dynamic or classical resolution
Jonathan Clayden, Przemyslaw M. Kubinski, Federica Sammiceli, Madeleine Helliwell and Louis Diorazio
Tetrahedron, 2004, 60, 4387-4397 [doi 10.1016/j.tet.2004.01.099]
97. Atropisomerism (Preface to Tetrahedron Symposium in print)
Jonathan Clayden
Tetrahedron 2004, 60, 4335
96. Nucleophilic addition to electron-rich heteroaromatics: dearomatising anionic cyclisations of pyrrolecarboxamides
Jonathan Clayden, Rachel Turnbull and Ivan Pinto
Org. Lett., 2004, 6, 609-611 [doi 10.1021/ol0364071]
95. Controlling chemoselectivity in the Lithiation of Substituted Aromatic Tertiary Amides
David R. Armstrong, Sally R. Boss, Jonathan Clayden, Robert Haigh, Basel A. Kirmani, David J. Linton, Paul Schooler and Andrew E. H. Wheatley
Angew. Chem. Int. Ed., 2004, 43, 2135-2138 [doi 10.1002/anie.200353324]
94. Fast racemisation and slow epimerisation of laterally lithiated amides: stereochemical evidence for the mechanism of inversion of amide-substituted benzyllithiums
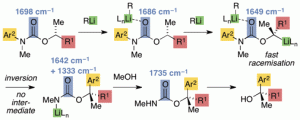
Jonathan Clayden, Christopher C. Stimson, Martine Keenan and Andrew E. H. Wheatley
Chem. Commun., 2004, 228-229 [doi 10.1039/b310963a]
93. Atropisomers and near-atropisomers: achieving stereoselectivity by exploiting the conformational preferences of aromatic amides
Jonathan Clayden
Chem. Commun., 2004, 127-135 [doi 10.1039/b307976g]